
So. You want to see a laser. That's why you're here, right? Well, I won't disappoint: laser fun will be had by all. But you have to promise me that you will at least try to learn something. Deal? There won't be a test (though there will be a poll), but if you don't learn something you will always look back on this day with longing and regret, wondering what might have been. Really. Oh, and for you smart-alecky scientists and enthusiasts who already know this stuff, shut-up and stop ruining it for everyone else. You know who you are.
I don't play around with lasers just for fun. Don't get me wrong, they are fun, but they can also be very useful (when they aren't exploding). So this series is not only going to cover some basic laser science, but will also explore my field of research: laser cooling and trapping. Wait! Don't leave! I'll be sure to keep it nice and easy. Plus, if you stay a certain someone may just burn something with his yellow laser, just for you. Alright then. Now put on your laser goggles and follow me over the jump!
Part I: What is that red spot doing on my pants, and why does it keep moving? (or How a laser works
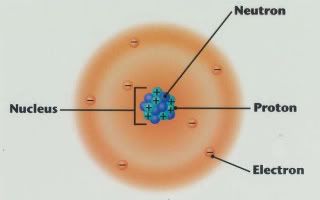
Energy levels, absorption, and emission
Atoms consist of small, negatively charged particles called electrons orbiting a relatively large, positively charged nucleus of protons (positive) and neutrons (neutral). Electrons are bound to atomic nuclei via the electric Coulomb force, much in the same way that the planets orbit the Sun because of gravity. But in the case of electrons and nuclei, because of quantum mechanics, the orbits can only have discrete, not continuous, energies (think the speed and size of the orbit).
These orbits can change in energy through emission and absorption of photons, which are little packets, or 'quanta', of light. The color of light is related to the energy of its constituent photons. Because energy is conserved, the change in energy of an atomic orbit has to equal the energy of the absorbed or emitted photon (you can't get something from nothing, after all).
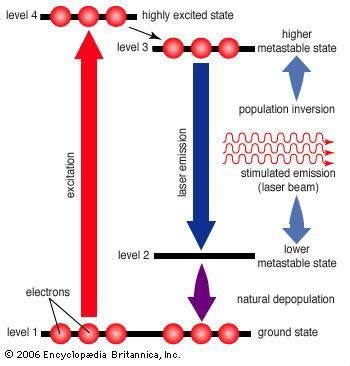
Laser transitions, the pump, and the gain medium
Lasers can be understood using a four level atomic model like the one shown to the right. The atoms start in level 1, and are excited to level 4 by an external energy source called a pump. The pump can be optical, electrical, chemical, or even nuclear, but has to supply enough energy to get many of the atoms into level 3 via level 4 (population inversion).
Once in level 4, the atom will quickly transition into level 3, or the higher 'metastable state'. This state is called 'metastable' because the atom spends much more time here than in level 4.
Under normal conditions the atom will fall spontaneously into level 2, which gives off a photon in a random direction. However, if somebody places the atoms in between two properly designed mirrors (a cavity), then some of those randomly emitted photons can bounce back through the atoms. This can cause another already excited atom to give off a photon in the same direction without destroying the original (stimulated emission). Thus, you get two photons for one. The atoms can now be called a gain medium, as the light amplifies while photons continue to bounce back and forth inside the cavity. (Important note: a gain medium doesn't have to be atomic. In fact, most aren't.) From level 2, some atoms will fall off back into the ground state to start the cycle anew. Assuming that all of the parameters are just right, the amplification is huge and the system begins to lase.

The output coupler and safety
It turns out that if we let a few of the laser photons 'leak' out, there are still plenty left inside the cavity to keep the process going. All lasers have one mirror that doesn't reflect all of the light incident on it, but lets a few percent through. This is called the output coupler, as it couples out the light. But don't let 'a few percent' fool you. Laser emission is dangerous; never, ever shoot a laser pointer into someone's eye. High power lasers should be left to professionals. (Hey, that's me!)
Lasers in the laboratory and a video quiz
OK, now the fun stuff I promised. Let me introduce you to my friend, the Coherent CR-699 Ring Laser:

Here is its gain medium:

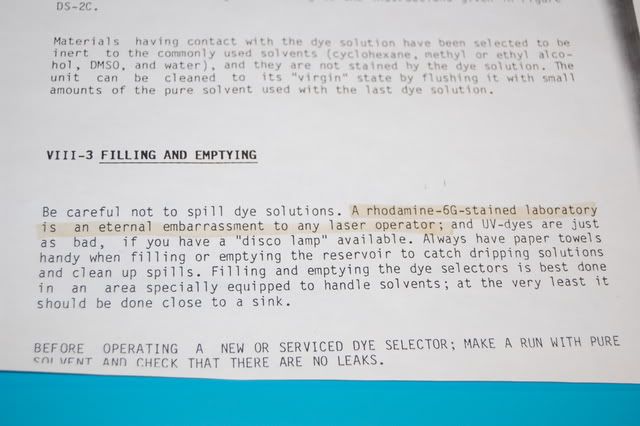
That's Rhodamine 6G dye mixed in ethylene glycol (like anti-freeze) being propelled across free space at a pressure of 60 psi. The dye is the worst part of working with this laser. A page in the operator's manual:
Our lab:



To be fair, that last spill was Rhodamine B, but you get the idea. Anyway, the 699 is pumped with (for now) a Spectra 171 large frame argon-ion laser:

In this case, the gain medium is actually a plasma that is created in a long tube and confined by a magnet. 30 amps at 500 volts (thats 15 kilowatts) are required to produce 7.5 watts of laser light. Talk about inefficient. These guys are no fun when they explode.
Now for the video quiz. First, a demonstration of the 699 output power:
That was me holding a card in front of about 1 watt of laser light. Burned pretty quickly, right? Remember how the output coupler only lets out a few percent of the total power? The beam inside the cavity is easily many tens of watts. So surely nobody would be crazy or stupid enough to put their hand inside the cavity, right? Well, guess again, unbelievers:
Unscathed. So, am I a magician, or was the video a hoax? Neither! If you were paying attention earlier you would have realized that if the photons can't bounce off of the cavity mirrors then there is no amplification, and thus, no laser.
Well, that's it for this part of the series. There is still much to explore in the cooling and trapping lab (a teaser: Laser Lightning in the Lab), but it will have to wait. Hope you enjoyed, and don't forget the poll!


