Crossposted at Politicook.net
There was a post the other day on Kos about a promising new source for hydrocarbon fuels from biomass. On further examination, biomass was only part of the feed, the rest being sugars and starches. The poster and a couple of others took offense when I pointed out that cellulose is quite different than starch.
I will use tonight's spot to explain. Sugars, starches, and cellulose belong to a large group of compounds called carbohydrates. This name is used because they all have the general formula (CH2O)n, where n can be from 3 up into the thousands.
Carbohydrates can consist of single subunits, or of two or more subunits joined together by elimination of a water molecule. The single subunit ones are called monosaccharides, two joined together are diasaccharides, and so on, with many subunits joined together are polysaccharides. There are only a few monosaccharides with n=4, and they are not often encountered. There are more with n=5, and two very important ones are ribose and deoxyribose, the building blocks of RNA and DNA. (Technically, deoxyribose is not a carbohydrate since it is an oxygen short of the general formula, hence the "deoxy" part.)
Monosaccharides with n=5 or greater have the interesting property of being able to exist either in a ring or in an open chain. Here are the representations for fructose (fruit sugar, n=6, and a component of sucrose, the diasaccharide that forms table sugar).
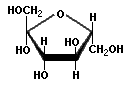 Fructose ring
Fructose ring 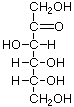 Fructose open chain
Fructose open chain
Because of different functional groups, the ring form and the open chain forms have different properties. This becomes important with more complex carbohydrates. The monosaccharides exist in water solution in equilibrium with each other, changing back and forth.
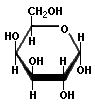
There are many n=6 sugars, and glucose is the other really important one. There is a bit of a difference from fructose, however. Glucose can cyclize in two ways, either with two —0H groups on opposite sides (the 1 and 4 positions) of the ring "down", or with one "up" and one "down". When they are both "down", it is called α-glucose, with one of each it is β-glucose. This becomes important later. Here are the representations of it:
 α-Glucose
α-Glucose 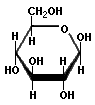 β-Glucose
β-Glucose 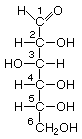 Open chain Glucose
Open chain Glucose
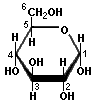
There is another monosaccharide that we commonly encounter combined with glucose in lactose, milk sugar. It is called galactose, and is represented by:
 Galactose
Galactose
Note that is just like glucose except for the way that the —0H groups are arranged at the 1 and 4 positions. Some other post I will discuss the subtle implications of stereochemistry, but not tonight.
Fructose is sweeter than glucose, and is metabolized somewhat differently, thus making is a part of diabetic diet. But be not deceived, it is still sugar and too much of it is bad for anyone. This brings up the topic of high fructose corn sweetener (HFCS). I will address it in a couple, after we discuss sucrose.
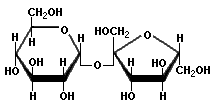
We do not encounter monosaccharides very often, except for fructose in the store and glucose in an IV at the hospital (the medical types refer to it as dextrose, but that is just a different name for α-glucose). The three sugars that we are most familiar with are sucrose (table sugar), lactose (milk sugar), and maltose (malt sugar). Sucrose is available in large quantities in bags, lactose is the sugar in milk and whey products, and maltose is the sugar in barley malt (we usually do not get it directly, but most flour contains malted barley flour, hence maltose).
These are all diasaccharides, formed from the condensation of two monosaccharides. Here are the representations for them:
 Sucrose
Sucrose 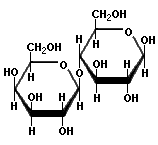 Lactose
Lactose
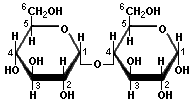 Maltose
Maltose
Sucrose is one glucose and one fructose joined in what is called a 1α→2 glycoside link. The 1 and 2 part mean that the 1 position of the first monosaccharide is joined to the 2 position of the second one. The α part means that the link is on the "bottom" of each unit, as represented by the diagram above. Maltose is formed from two glucose units in the same α spatial arrangement. Lactose is different. It is one galactose and one glucose joined by a 1β→4 glycoside link. The 1 and 4 mean that the 1 position of the first unit and the 4 position of the second unit are involved, but the β part means that the "top" of the galactose is connected with the "bottom" of the glucose. I know that this seems complicated, but it is absolutely critical to understand the difference between α and β links to understand the differences between starch and cellulose, and indeed other materials as well.
This is bit oversimplified, but illustrates the concept. The fundamental difference is that the geometries are quite different, and those geometries results in different chemical properties. It turns out that there are lots of enzyme systems that can attack α links, but not so many than can attack β ones. The α ones are also more sensitive to heat, particularly moist heat, and also to acid attack. That brings us now to high fructose corn sweetener. Well, not quite yet. First we have to consider starch, but we are getting there. There are other possibilities for bonding two monosaccharides together, with the 1 and 6 positions being common. Some materials have both 1,4 and 1,6 links.
Starch is a long chain system of glucose. It comes in two basic forms, amylose and amylopectin. Amylose is straight chained, and arranges itself into a nice helical arrangement when dispersed in cold water, to which it hydrogen bonds. Amylopectin is highly branched, and does not have much to do with water except to fall to the bottom. Here are the diagrams of them:
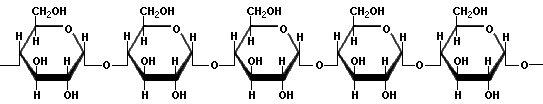 Amylose
Amylose
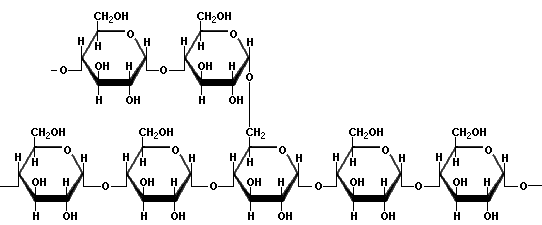 Amylopectin
Amylopectin
Although it is hard to see from the diagram, the link in amylopectin has the geometry of an α link, so the side branches are termed 1α→6 glycosidic links. Anyone who has had a busy Thanksgiving dinner preparation time is familiar with the hazards of amylopectin. You are getting the turkey out, making sure the roll go in, hoping the sport fans will help rather than just eat, and you stirred the cornstarch into the cold water for the giblet gravy. Then something happens to distract you, and when you come back to finish the gravy in five minutes, your spoon is stuck in the cornstarch. The amylopectin has settled to the bottom of the cup or bowl like concrete. The amylose is still suspended, but you strain your arm to get everything back in the cold water. By the way, amylose contains anywhere from around 20 to 20,000 glucose units, and amyopectin from thousands up to a couple of millions of glucose units. Each 1,6 sidechain may have a couple of dozen units, but those can branch as well, making for a very complex structure.
Moist amylopectin displays some interesting physical properties, and it is easy to make. Just take a cup of cornstarch and suspend in in about a quart of water. If you want to keep it for any length of time, put a couple of tablespoons of borax in it to keep the mold and bacteria from eating it fast. Stir it all up, and then let it settle. Do this in a wide bowl, because it is sort of hard to get out of a jar. Put it on a surface that moisture will not damage and try some of these.
Slowly shape it sort of like clay. It will not take a definite shape like clay will, but you can make a ball of sorts out of it. Then slowly push down from the top and it will flatten easily. Now make a new ball and punch it down with your fist. It will hardly distort at all. Perhaps a reader would elaborate. Now back to starch.
Starch is entirely glucose in 1,4 and 1,6 (all α) links. We humans can digest α links, so starch is a good food source. When starch is treated with certain enzymes or acids, it adds water to the links and becomes essentially pure glucose. That is Karo syrup, the baker's delight. There is a problem with glucose, though. It is only 74% as sweet as cane sugar (sucrose). But it makes nice pecan pies.
OK, now for high fructose corn sweetener. It is Karo that has been treated with enzymes to convert some or nearly all of the glucose to fructose. Why do that? Fructose is almost twice as sweet as sucrose, and nearly three times as sweet as glucose. Converting a less sweet sugar into a more sweet one allows less to be used in, say, soft drinks, to allow for the same level of sweetness. That was the entire idea about the "New Coke" back in the early 1980's. Cheaper to use. But there is more.
No one thought that I would leave politics out, did they? A huge driving force towards the use of HFCS is the United States tariff on sugar. As of Friday, according to Agricultureonline.com, the price of sugar on the world market is 11.65 cents per pound, but the US price is 22.95 cents per pound, due to the tariff. Close to double. No wonder Life Savers are now made in Canada (finished products are not covered by the tariff, just the sugar). So if you are a soft drink maker, and use sugar in multi ton quantities, why not use something cheaper? Now you know. Not only is HFCS not covered under the treaty, it is almost twice as sweet as sugar. So your cost is cut by almost 75%. That is a no brainer for a Yale MBA, well for most of them, unfortunately.
The question remains: is HFCS bad for you? No, I my opinion, with this caveat. A 12 ounce can of the most popular cola has 140 calories, and 39 grams of carbohydrate. All of the calories are from carbohydrate. Many people drink the equivalent of three or even more cans a day, for, say 420 calories with no nutrients other than carbohydrate. In comparison, my bottle of generic 100% juice of the cranberry variety has, for 8 ounces, the same 140 calories, 35 grams of carbohydrate, and 4% of the RDA of potassium and 130% of the RDA of Vitamin C.
My point is that HFCS is not the enemy, it is calories with no benefit. Most fruit has the same ratio of fructose to glucose as modern soft drinks, but many nutrients in addition. I suggest that you get your calories from something that gives you vitamins and minerals as well. Some people say that the sucrose sweetened soft drinks have a slightly different flavor and mouth feel than the HFCS sweetened ones. I do not know, since I drink soft drinks only rarely. I am told by those who claim to know that Coca-cola made in Mexico is "better" than the US product because it is still made with sucrose.
Now to the big one: cellulose. That is richest source of sugar that exists in our world. But it is hard to process. Why? Unlike starch, the links between the pure glucose units are 1β→4 links, and they are much, much more difficult to break. Acid will not do it except under extreme conditions, and even then one ends up with cellibiose, which is two glucose molecules joined by a 1β→4 link. Modern smokeless powder is made by boiling cellulose with concentrated nitric and sulfuric acids, and all that they do is replace some of the side chains, not break the long polymer. Here is a diagram of cellulose:
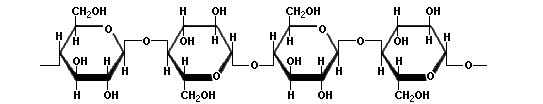 Cellulose
Cellulose
The notable features of cellulose are that it forms very long, linear chains with no branching, the links are the difficult to cleave 1β→4 links, and those long polymers hydrogen bond with each other like crazy, resulting in an extremely inert material.
Enzymes will not do it, at least higher animal ones. Some bacteria have figured it out, so termites and cattle can digest cellulose anerobically with their assistance. This brings us back to lactose. Remember, it is a galactose and a glucose joined with a 1β→4 link, and those are hard to break. Many people have the enzyme needed, but many others do not. Stomach acid will cleave 1α→4 links, but not 1β→4 ones. Only monosaccharides can be absorbed and metabolized, so if you do not have lactase, the enzyme specific for cleaving this link, you are out of luck. The lactose is not absorbed, and enters the large intestine unchanged. It then acts as an osmotic laxative, absorbing water and causing the usual effects of an osmotic laxative. Other osmotic laxatives include Epsom salt and the dreaded magnesium citrate (the little bottle that you drink before undergoing a diagnostic procedure).
Now, more about cellulose. It is an excellent fuel, because the glucose subunits have a relatively high energy content and at combustion temperatures the 1β→4 links are easily split. However, cellulose is a solid (anywhere from a few hundred to many thousands of glucose subunits comprise it), so it is sort of hard to pump. It works well in the fireplace, however. If only there were a way to convert it to a more usable form. That is the promise of the so-called biomass fuel process.
It is possible to cleave those links given enough brute force, and I do mean brute force. So much force that most processes to date are not economical in converting cellulose to liquid fuels. Some processes are being developed based on enzymes such as those found in termite gut, but even at $100 per barrel, oil is still cheaper. Other processes are being developed that use catalysts and heat and pressure, but they still require input of mono or disaccharides to make the process work, and that means using food as at least part of the starting materials. It is just hard to depolymerize cellulose!
Cellulose is all around us. Wood is predominately cellulose with lignin to bind the fibers together. Lignin is a complex material composed of long polymers of highly substituted alcohols, and forms the "glue" that holds the cellulose chains together in wood, making it rigid and strong. With all the talk about modern composite structural materials (like fiberglass or carbon fiber composites), wood is the prototype. All that we have done is mimic nature.
The most pure form of cellulose that we encounter in everyday life is cotton, especially in the form of cotton wool, like the familiar cosmetic cotton balls. This is almost pure cellulose, with a little water hydrogen bonded to it. If you were to comb out the fibers and put glue on them and then compress them, the resulting product would be very much like wood.
Cotton fibers are among the longest cellulose chains, and that makes them well suited for textiles, since longer fibers are easier to spin into cloth than shorter ones are, and thinner threads are feasible when only a few long fibers are spun. That is why textiles are not made with wood pulp. Those fibers are too short to make good cloth, but they are long enough to mesh together to make paper. There are a few exceptions, where carefully selected wood and careful processes are utilized to produce textiles, but that is not very common.
US currency is predominately cotton fiber paper, hence the long wear and good texture. If wood pulp were used, the money would crease and tear mush more readily than it does.
Other sources of textile cellulose include linen, grown in temperate climates in the form of the flax plant. It produces a very nice fiber, but is expensive when compared to cotton, and lots of processing has to be done to separate the fibers from the plant. In cotton, the fiber is in the seed pod, and all that has to be done is comb out the seeds, the process known as ginning. In linen, the fibers are in the stem of the plant, and they have to be separated by a wet process, called retting, that essentially rots out the lignin and allows the fibers to be recovered.
Hemp also provides a good quality cellulose for textile and paper uses, but there are some legal issues involved. It has to be retted as well, since the fibers are in the stems. At one time hemp was heavily used for paper in the United States, and that brings up another political issue. I do not feel like taking that one on tonight.
There are many, many other sources for cellulose, and lots of them are waste products. For instance, most sugar refineries are run off of the heat produced when the sugarcane is burnt after the sugar is extracted. In Hawaii, at least one sugar refinery produces so much excess steam that it generates electricity with it and sells it to the grid.
I am optimistic that cellulose, particularly waste cellulose will be used someday for energy applications, but the current technology is just not developed to the point that we can pump it into our cars. Ideally, we would be able to extract the hydrogen from it, leaving the carbon behind for disposal (it is easier to dispose of solid carbon then it is carbon dioxide). We actually do than on a limited scale in the charcoal making process, capturing the volatiles. That does not separate the carbon completely, since some of the volatiles contain carbon. The think is, we turn around and burn the charcoal as well since it is such a good fuel.
Herein lies the problem with energy: no other common material produces nearly as much heat as carbon when it is burnt except for hydrogen on a mass basis. Hydrogen produces a little over four times the energy on a gram per gram basis, but that hydrogen has to come from somewhere, and that somewhere is generally water. Well, then we have to put that energy into water to split out the hydrogen, since water is already burnt. It is the same situation as putting energy into carbon dioxide to split out the carbon.
That the rub with hydrogen. Hydrogen produced by the electrolysis of water is fine, but it is extremely expensive unless electricity is cheap. What is the cheapest source of electricity currently? Except for a few locations, like hydropower areas, it is coal fired power plants. That does not help! Nuclear or solar could produce it cleanly, but hardly cheaply.
Hydrogen is used industrially in multi-ton amounts for everything from the petroleum industry, the fertilizer industry (to make ammonia, the starting material for other products), and many other industries. Where does it come from? The refining industry produces some of its own, but almost all of the rest is produced by blowing steam through incandescent coal. In other words, coal is burned in air (producing carbon dioxide), then the air supply is cut off and steam is blown through. The products are predominately hydrogen, carbon monoxide, a little nitrogen (left over from the air and also present in small amounts in coal), and carbon dioxide.
It takes over four grams of coal burning to carbon dioxide to produce one gram of hydrogen this way. Actually, it takes more because we had to get the coal burning before we added the steam, and the reaction producing hydrogen cools off the coal, and so it has to be cut off and air started again to heat the coal back up to make the reaction go again. Thus, making hydrogen except by using electrolysis based on a carbonless electrical supply is actually much worse than just burning the carbon source directly for power. Conservation of energy is a hard master.
Well, I got a little off topic there at the end, but that was an important point to make since so many are confused about using hydrogen as a fuel. I have no problem with is as a fuel, just the way we have to get it. Questions, comments, flames, and elaboration about these or any other science and technology subjects are, as always, welcome.
Warmest regards,
Doc
Update: this one did not go over as well as I had had thought that it might have. No matter! Already working on the one for next week, about forensic science, with a safe, cheap, and exciting do it at home project! I suppose that many are out having the last fun for the summer, and others are worried about the economy. I wanted to post a diary about coins yesterday, but formatting problems prevented it. I think that I have overcome them now, so those of you who like coins, watch for my next installment coming around about 8:00 PM tomorrow, with pretty pictures! And I will be around for a little while yet, so keep those questions and comments coming!
Warmest regards,
Doc


