Crossposted at Politicook.net
This is the second stab at mass spectrometry. The first one just got too wankish for anyone to want to read, so I threw it out and started over. Instead of spending a whole lot of time explaining how it works, I'll just do a brief description the hows. I will be happy to address more detail in comments.
We found out last week that chromatography is a powerful method for separating complex mixtures into individual components, and that we can make a pretty good guess as to what those components are by comparing retention times with known standards, but that retention times are not necessarily unique to a particular compound.
What we need is a detector that can identify a molecule without and uncertainty. Mass spectrometry is this detector.
There are many different kinds of mass spectrometers, but they all operate in the same way. There are four processes involved. The first is sample introduction, and there are interfaces for both GC and HPLC that serve that purpose. It is easy with GC, and much harder with HPLC, but it can be done.
The second step is ionization of the molecules. Once ionized, they can be accelerated and focused electromagnetically, just like the electrons in a CRT. In addition, the energy from ionization often causes molecules to fragment, and fragmentation patterns are a fingerprint of a specific molecule. There are several ways to do this, the most common hitting molecules with a stream of 70 volt electrons. This knocks one or more electrons off, leaving ions with a positive charge.
The third step is to sort out the ions by mass (technically, it is the mass to charge ratio, but we will assume that our ions are singly charged). There are several ways to do that, but most benchtop instruments use a quadrupole mass analyzer because they are relatively inexpensive, easy to operate, and easy to maintain. In the quad there are four conductive rods arranged in a square. Two opposing rods get a positive electrical charge, the other two a negative one. A high frequency AC sweep is impressed on top of the static charge. Depending on the frequency of the sweep, only one mass will have a stable path through the quads, and all other masses hit the quads and are stopped. This sweep can be done fast, so many different ions can be analyzed in a second.
The forth step is detection. Most modern instruments use an electron multiplier tube that reacts to an ion hitting the surface of it with a cascade of electrons big enough to produce a measurable current. This current is amplified and than recorded by computer. The electronics coordinate the mass analyzer with the detector, so the computer "knows" when each mass is hitting the tube.
What emerges is a picture of the fragmentation pattern of the molecule. Here are pictures of the mass spectra of three different compounds with similar chemical formulae but different structures:
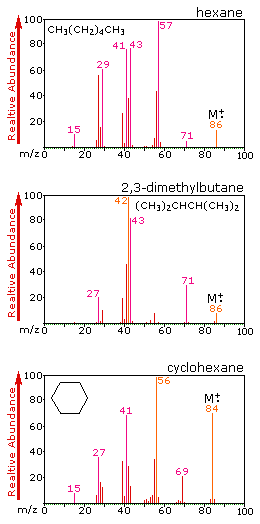
The top two have identical formulae. Note the difference in the mass spectra in them. There are rules that people use to determine which is which if no standards are available.
But it gets better. There are computer databases available with tens or hundreds of thousands of mass spectra that can be searched for matches, thus facilitating figuring out what is what.
There is still the problem with the potential for chromatographic coelution (two or more components with the same retention time). Mass spec comes to the rescue then, too, because let us say that the top two compounds coelute in your setup. Simply by checking to see whether or not the mass spectrum has a significant peak at 57 tells you which one you have.
The spectra just shown are called full spectra, because the entire range from 0 to 100 was scanned. It is possible to set an instrument up such that only a few ions are detected. This is called selective ion monitoring and is great when you have analyses of similar materials to do over and over, because it gives better sensitivity. That is because you are looking only for ions of interest at a specific retention time, to more time is spent by the instrument collecting those and not ones that do not matter. You usually pick out the biggest ones, but often you use smaller ones too, because they are more or less unique for your material. Commonly, three to five are looked for in this technique.
So now we can separate out complex mixtures and then look at each component in detail. It is extraordinarily unlikely for two different compounds to have the same retention time and the same mass spectrum. That is why GCMS is used in forensics, because, if samples are handled properly, GCMS identification is airtight in court.
Drugs testing usually consists of two phases after the specimen is collected. First is a fast screening that does not cost much nor take much time. Unfortunately, there is a finite probability that a false positive will occur. If a screening sample shows positive, a split of that sample is sent for GCMS analysis. If it comes in positive there, you are looking for a new job. There are very strict guidelines for judging a sample positive, such as minimum quantities, quality of the chromatogram and the mass spectrum, blank and control sample results, and chain of custody of the sample.
When I worked where there was random drugs testing, I always requested that they take two samples and put on the seals, then keep one until the results came back. I knew that I was clean, but as a professional scientist I also know that people in laboratories make mistakes, and sample preparation personnel are amongst the lowest paid technicians. I have prepared and supervised the preparation of samples long enough to know that sometimes labels get switched. That second specimen was my hedge against that. I strongly recommend that you do the same if you work where they do random drugs testing as well.
Whilst we are on the subject of drugs testing, please allow me the soapbox for a minute. I have fundamental issues with it on personal liberty grounds. I think that I know what Franklin and Jefferson would have had to say about it as well. But that is the reality in the Land of the Free. If you are clean, do NOT feel confident that the lab will say so.
Cannabis is particularly a problem, for a couple of reasons. First, being fat soluble, cannabinoids stay in the body for a long time, thus making it hard to get away with a joint or two whilst on vacation, for instance. Two weeks for occasional use, and a month or more for regular use is not uncommon to end up positive. Most other drugs remain for only a couple of days before they are eliminated since they are water soluble.
I intentionally kept this relatively brief, because it is easy to get really detailed and boring. As I said earlier, any questions for more detail are welcome in the comments.
UPDATE: I found a problem with my picture. Fixed now.
Warmest regards,
Doc


