Like a geologic tombstone, the red clay layer marks the major extinction event that happened the last time rapid atmospheric CO2 increases caused the oceans to acidify, 55 million years ago.

Sediment core spanning the Paleocene-Eocene Thermal maximum.
The mid-ocean became so acidic that calcium carbonate, the white material in the sediment above and below the clay layer dissolved in sea water. The calcium carbonate came largely from the hard body parts of marine organisms. The red clay layer signifies an acidic environment when many marine species went extinct.
A new study shows the oceans are acidifying ten times faster than the last major marine extinction 55 million years ago.
As levels of atmospheric CO2 increase, the oceans acidify. It's as certain as death and taxes. About one third of all carbon dioxide burned ends up dissolved in sea water as carbonic acid. The weathering of rock by rain, acidified by CO2, takes hundreds of thousands of years to neutralize ocean acidification. While journalists in major newspapers proof read the details of the IPCC report on climate change, reporting on the latest controversy manufactured by "climate skeptics", two key reports on the acidification of the oceans have gone largely unnoticed in the American media.
Acidification of the ocean by anthropogenic CO2 is undeniable.
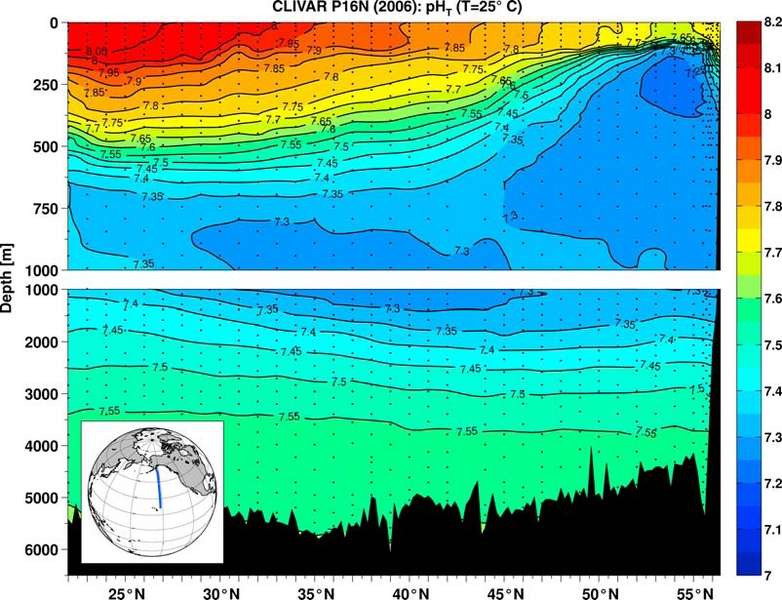
[1] Global ocean acidification is a prominent, inexorable change associated with rising levels of atmospheric CO2. Here we present the first basin-wide direct observations of recently declining pH, along with estimates of anthropogenic and non-anthropogenic contributions to that signal. Along 152°W in the North Pacific Ocean (22–56°N), pH changes between 1991 and 2006 were essentially zero below about 800 m depth. However, in the upper 500 m, significant pH changes, as large as −0.06, were observed. Anthropogenic and non-anthropogenic contributions over the upper 800 m are estimated to be of similar magnitude.
In the surface mixed layer (depths to ~100 m), the extent of pH change is consistent with that expected under conditions of seawater/atmosphere equilibration, with an average rate of change of −0.0017/yr. Future mixed layer changes can be expected to closely mirror changes in atmospheric CO2, with surface seawater pH continuing to fall as atmospheric CO2 rises.
The pH of the Pacific ocean in 2006 on a transect along 152W from Hawaii to the Aleutian Islands.
Oxidation of organic matter sinking into the mid-levels of the ocean creates a low oxygen zone with a temperature adjusted pH as low as 7.25. Upwelling of the acidic, low-oxygen mid-ocean water along the Aleutians and the coast of the Pacific northwest has created a growing dead zone in the Pacific.
Pacific Ocean pH change from 1991 to 2006

Rapid acidification was observed over 15 years along the transect.
Cyclic changes in ocean currents and ocean mixing caused some of the pH changes from 1991-2006. A slow down in mixing in the decade of the '90s allowed more CO2 build up than normal from oxidized organic matter. The investigators subtracted pH changes caused by the natural variability of ocean processes to determine the anthropogenic component of the acidification of the north Pacific.
About half of the decrease in pH was attributed to anthropogenic carbon and half was attributed to ocean circulation changes. Over time, as anthropogenic CO2 emissions continue and natural variability averages out anthropogenic carbon will be the dominant cause of ocean acidification. Increasing acidity will be incompatible with the survival of pH sensitive marine organisms at the bottom of the food chain eaten by salmon and other key fish in the north Pacific and north American ecosystems.
Nevertheless, the section-wide (22°–56°N) average of ΔpHant between 800 m and the mixed layer amounts to −0.011 (±0.001). This change corresponds to 48% (±10%) of the total pH change.
[18] On multidecadal time scales, pH changes attributable to anthropogenic CO2 can be expected to eventually dominate the signal. CO2 produced by respiration in the subsurface ocean (diagnosed from O2 measurements) can be expected to show positive and negative variations [Deutsch et al., 2006; Whitney et al., 2007; Mecking et al., 2008]. In contrast, anthropogenic CO2 in seawater should steadily increase as atmospheric levels continue to rise. By the end of this century, distinctions between total pH change (Figure 2) and pH change attributable to anthropogenic CO2 (Figure 4) will become increasingly subtle.
[19] One outcome of the observed widespread pH declines (Figures 2 and 4) is an increasingly inhospitable environment for calcifying marine plankton, such as pteropods, forams, and coccolithophorids. Decreasing pH translates directly to decreasing carbonate saturation states. The physiological status of these pelagic calcifying organisms currently abundant in the North Pacific is therefore likely to be adversely impacted [Feely et al., 2004, 2008; Fabry et al., 2008]. Many other chemical, physiological, and ecological processes are affected as well (e.g., trace metal speciation, photosynthesis, nitrogen fixation), to an extent that is not well characterized [Doney et al., 2009].
[20] The observed near-surface pH changes (Figure 2) are in good agreement with predictions based on CO2 system thermodynamics, consistent with attribution of most of this change to anthropogenic acidification (Figure 4) and with observations elsewhere. Average ΔpHm along 152°W in the zone shallower than the winter mixed layer is −0.026, equivalent to an average annual pH change rate of −0.0017. For seawater fully equilibrated with the atmosphere in 1991 and 2006, a ΔpH of approximately −0.025 would be expected. A directly measured surface-water decline of −0.0017 y−1 (pHT, 25°C, 1995–2004) has also been reported at a time-series station in the subtropical North Atlantic [González-Dávila et al., 2007]. Rates of change consistent with long-term air/sea equilibration have been reported for other CO2 system parameters in the North Pacific [Takahashi et al., 2006; Dore et al., 2009] and North Atlantic [Bates, 2007; González-Dávila et al., 2007].
Pacific Ocean pH change caused by anthropogenic CO2 emissions

The modeling study assessed the present rates of ocean acidification and the future with continued anthropogenic CO2 emissions. The scientists found that continued anthropogenic CO2 emissions will cause the acidity of the ocean to increase to the point that the survival of plankton, critical to the marine ecosystem and a major source of the earth's oxygen, will be threatened.
Increasing concentrations of carbon dioxide in sea water are driving a progressive acidification of the ocean1. Although the associated changes in the carbonate chemistry of surface and deep waters may adversely affect marine calcifying organisms current experiments do not always produce consistent results for a given species. Ocean sediments record past biological responses to transient greenhouse warming and ocean acidification.
During the Palaeocene–Eocene thermal maximum, for example, the biodiversity of benthic calcifying organisms decreased markedly, whereas extinctions of surface dwellers were very limited. Here we use the Earth system model GENIE-1 to simulate and compare directly past and present environmental changes in the marine realm. In our simulation of future ocean conditions, we find an undersaturation with respect to carbonate in the deep ocean that exceeds that experienced during the Palaeocene–Eocene thermal maximum and could endanger calcifying organisms. Furthermore, our simulations show higher rates of environmental change at the surface for the future than the Palaeocene–Eocene thermal maximum, which could potentially challenge the ability of plankton to adapt.


