I think that "Science" will one day 'save the day,' with respect to reversing the uncontrolled Planetary CO2 Experiment we are currently conducting. That is of course, if we don't first send "Science" -- and all the innovative minds behind it -- to the dustbins of history. To a the circular file labeled: "If Only ..."
Far too often however, even in the insular world of "Science" (the pursuit of natural and technological knowledge) and the sometimes dogged pursuit of new promising ideas, it ultimately comes down to the costs (and the potential profits, too).
Case in point, witness this epic battle between the daring innovators and the stodgy naysayers. Much hangs in the balance, depending on who ultimately wins the battle of public opinion (and the investment dollars, too).
My money's on the innovators ... in the long run. They're kind why we've gotten as far as we have.
This Climate Fix Might Be Decades Ahead Of Its Time (with Audio)
by Richard Harris, NPR, OPB -- June 27, 2013
Every year, people add 30 billion tons of carbon dioxide to the air, mostly by burning fossil fuels. That's contributing to climate change. A few scientists have been dreaming about ways to pull some of that CO2 out of the air, but face stiff skepticism and major hurdles. This is the story of one scientist who's pressing ahead.
Peter Eisenberger is a distinguished professor of earth and environmental sciences at Columbia University. Earlier in his career, he ran the university's famed Lamont Doherty Earth Observatory, and founded Columbia's Earth Institute. He was never one of those scientists who tinkered into the night on inventions. But he realized he didn't need to be.
"If you looked at knowledge as a commodity, we had generated this enormous amount of knowledge and we hadn't even begun to think of the many ways we could apply it," Eisenberger says. He decided he'd settle on a problem he wanted to solve, and then dive into the pool of knowledge for existing technologies that could help him.
He started looking for a way to pull carbon dioxide right out of the air. "And it turned out the best device already exists," he says. "It's called a monolith. That is the same type of instrument that's in the catalytic converter in your car. It cleans up your exhaust."
[...]
"Monolith"?
What is a Monolith?
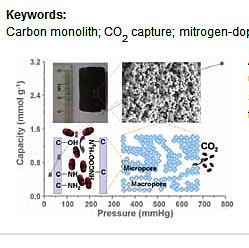
Carbon nanotube modified carbon composite monoliths as superior adsorbents for carbon dioxide capture
Yonggang Jin, Stephen C. Hawkins, Chi P. Huynh and Shi Su*
Energy Environ. Sci., 2013, Advance Article
DOI: 10.1039/C3EE24441E
First published online 01 Mar 2013
Carbon composite monoliths were prepared from a commercial phenolic resin mixed with just 1 wt% of carbon nanotubes (CNTs) followed by carbonization and physical activation with CO2. The products possess a hierarchical macroporous–microporous structure and superior CO2 adsorption properties. In particular, they show the top-ranked CO2 capacity (52 mg CO2 per g adsorbent at 25 °C and 114 mmHg) under low CO2 partial pressures, which is of more relevance for flue gas applications. This matches or exceeds those of carbons produced by complex chemical activation and functionalization. Our study demonstrates an effective way to create narrow micropores through structural modification of carbon composites by CNTs.
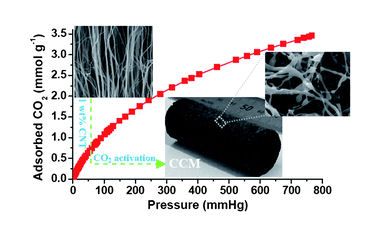
Rapid Synthesis of Nitrogen-Doped Porous Carbon Monolith for CO2 Capture
Guang-Ping Hao, Wen-Cui Li, Dan Qian, An-Hui Lu*
Article first published online: 12 FEB 2010
DOI: 10.1002/adma.200903765
A polymer gel derived from resorcinol and formaldehyde can be prepared in around 5 min using lysine as polymerization catalyst and nitrogen source. After pyrolysis of this polymer gel, a new type of nitrogen-containing carbon monolith was obtained (see figure), which exhibits the highest CO2-adsorption capacity to date of up to 3.13 mmol g[−1] at room temperature.
[... continuing toward the end of the NPR report: ]
Researchers currently working on carbon dioxide capture technologies say the American Physical Society critique has made it much harder for them to raise money. Klaus Lackner, at Columbia University says he was turned down for a government grant. David Keith, at Harvard and the University of Calgary, says he struggled to get funding for his small company.
"It's a very powerful report from a very credible group of people and it may well help to kill us and other efforts," Keith says.
Proponents of air-capture technologies say some of the panel's conclusions are just plain wrong -- especially the estimated cost of $600 per ton.
"We have had third party reports, independent people, evaluating our technology, and it's under $50 a ton," Eisenberger says. He hasn't actually demonstrated that cost yet, and he agrees that nobody should take his word for it. But he's stopped arguing with his critics.
"I'm just going to go do it," he says. "And doing it or not -- that's the answer."
Pursuing a big idea takes some hard-headedness, and thick skin.
[...]
As another 'boldly-go-where-few-have-gone' innovator -- he's going to need it (a thick skin).
Here's what the Society from 1899 has to say about Peter Eisenberger's new-found "technological optimism" ... with respect to simply one-day absorbing the excess CO2 out of the air ...
Direct Air Capture of CO2 with Chemicals (pdf)
A Technology Assessment for the APS
Panel on Public Affairs
The American Physical Society -- June 1, 2011
Executive Summary
This report explores direct air capture (DAC) of carbon dioxide (CO2) from the atmosphere with chemicals. DAC involves a system in which ambient air flows over a chemical sorbent that selectively removes the CO2. The CO2 is then released as a concentrated stream for disposal or reuse, while the sorbent is regenerated and the CO2-depleted air is returned to the atmosphere. To guide the reader to an understanding of the factors affecting costs, a benchmark system is introduced that could be built today. With optimistic assumptions about some important technical parameters, the cost of this system is estimated to be of the order of $600 or more per metric ton of CO2. Significant uncertainties in the process parameters result in a wide, asymmetric range associated with this estimate, with higher values being more likely than lower ones. Thus, DAC is not currently an economically viable approach to mitigating climate change. Any commercially interesting DAC system would require significantly lower avoided CO2 costs, and thus would likely have a design very different from the benchmark system investigated in this report. This report identifies some of the key issues that need to be addressed in alternative designs.
[...]
DAC costing
To evaluate a large DAC facility that could conceivably be built today, this report uses a simplified costing methodology applied in industry to early-stage projects. The benchmark DAC system is assumed to have a capacity of 1 MtCO2/yr and to absorb CO2 by passing air over a solution of sodium hydroxide in a counter-current, closed system. The sodium hydroxide solution containing sodium carbonate is then cross-reacted with calcium hydroxide to form calcium carbonate as a precipitate. The solid calcium carbonate is decomposed in a natural-gas-fueled, oxygen-fired kiln, with capture of the released CO2. The capital cost is estimated to be 2.2 billion dollars, a normalized cost of $2200/(tCO2/yr). Capital recovery contributes 60% of the $600/tCO2 estimated avoided cost.
[...]
For the sake of comparison, using the same methodology the avoided cost for “post-combustion capture” (PCC) of CO2 from the flue gas of a reference coal power plant is estimated. In the reference PCC system the CO2 is 300 times more concentrated and the CO2 removal rate is about three times larger than in for the benchmark DAC system. Relative to the benchmark DAC system, the normalized capital cost for the reference PCC system is estimated to be $180/(tCO2/yr), twelve times smaller, and the total avoided cost for capture is estimated to be about $80/tCO2, about eight times smaller. Since the total cost includes both operating and capital costs, evidently the operating cost ratio for the two systems is less disadvantageous to the benchmark DAC system than the capital cost ratio. One reason the ratio of operating costs is smaller than the ratio of capital costs is that the assumed energy requirements for the DAC system are optimistic. For example, DAC electricity demand includes fan power to move the air, which is proportional to the pressure drop through the contactor, and the pressure drop assumed in the cost calculation is at the very low end of a credible range for the benchmark system.
The capacity to estimate future DAC costs is limited. Costs could fall as a result of technological learning and with the introduction of fundamentally new ideas. On the other hand, industry experience suggests that cost estimates for any system rise after the completion of pilot plant operations, when the necessary compromises in materials choices, process conditions, component efficiencies, and component lifetimes are taken into account.
I searched this APS critique to see if they were current with the latest Materials research, with respect to Carbon adsorption processes being pursued by innovators like Eisenberger.
A search for "monolith" -- a key concept in Eisenberger's research -- revealed that the American Physical Society referenced the idea twice -- in the footnotes:
73. Hao, G., et al., Rapid Synthesis of Nitrogen-Doped Porous Carbon Monolith for CO2 Capture. Advanced Materials, 2010. 22(7): p. 853-857.
76. Chen, C., et al., Amine-impregnated silica monolith with a hierarchical pore structure: enhancement of CO2 capture capacity. Chem. Commun. , 2009. 24: p. 3627-3629.
Here's is the text which references Footnote [76], which itself references "monolith;" text from pg 60:
The water vapor in this example helps displace the CO2 and then sweep the CO2 away from the base site, preventing re-binding. In this example, water serves two roles by displacing the CO2 and carrying it away. This case is distinct from a situation where an inert gas simply sweeps away a small amount of desorbed CO2 and continues to sweep away the CO2 as it desorbs. The approach using an inert sweep gas results in a very large excess of sweep gas in the effluent relative to the amount of CO2 [76].
Among the step 5 questions/challenges are:
• In air capture systems where a second material (e.g., CaCO3) is required to concentrate the CO2-carrying entity prior to CO2 release, can materials be developed that lower the temperature of CO2 release?
• Can catalysts be developed to reduce the activation energy and lower the temperatures needed for CO2-release processes?
• Can liquid absorbents be designed that reduce or eliminate evaporative or carry-over losses that may occur, especially during the CO2 desorption step?
• Can organic liquid absorbents or organic functionalized CO2-capture adsorbents be developed with increased resistance to oxidation and hydrolysis and/or with improved thermal stability during CO2 release?
• When displacement by another molecule is used for CO2 release, can lower-volatility molecules be developed which result in CO2 being a larger fraction of the effluent, and/or which require less energy to regenerate the sorbent than when using water?
I could not identify where Footnote [73], was referenced in the APS critique. I'm guessing it's somehow "implied."
Additional keyword searches within the APS (old-school) critique, resulted in 0 search hits:
nanotubes
nanotech
nano-tech
carbon tubes
CO2-adsorption
catalytic converter
doping
The keyword search for "resin" however had a hit on pg 49:
Binding of CO2 to an adsorbent occurs at basic sites in, or attached to, the surface of a fixed solid structure, or to counter-anions (e.g., OH-) associated with a positively charged functional group (e.g. alkyl ammonium) that is incorporated into the backbone of a solid material such as a polymer resin (as in ion exchange resins used in water treatment) or metal oxide [55]. As with absorption by flowing liquids, efficient adsorption onto fixed solids requires a high-surface-area interface and the effective use of a high fraction of the binding sites. High surface area is achieved by using solid support materials which contain small pores, often referred to as micropores, whose diameters range from nanometers to micrometers. The basic sites that bind CO2 are then added to the surface of the pores.
Such microporous solid structures result in effective surface areas of 10s to 100s of square meters per gram of material. This property translates into tens to hundreds of millions of square meters of surface area per cubic meter, many thousands of times larger than the corresponding values for the absorption column and cooling tower packing materials reported above. However, because of the surface tension of water and the presence of an average of ~0.5% water in ambient air, water vapor condenses into the micropores of these high surface area materials. These materials, as currently available, may contain as much as 50% water by weight at ambient conditions. As a result, adsorption chemistry within the solid pores often parallels aqueous solution chemistry. For DAC applications using such materials, transport of CO2 to binding sites after crossing the interface (Step 3) may involve diffusion through a liquid. Additionally, although water is a weaker acid than CO2, the presence of an excess of water near the binding sites may interfere with CO2 binding at the basic sites.
"resin" was mentioned again on pg 55:
There is considerable interest in using solid adsorbents for capture of CO2 from air. A 1969 patent proposed a scheme to capture the excess CO2 in the air in a submarine [67]. It reported that over 99% of the CO2 in the air could be captured in a strong-base ion exchange resin, that the CO2 could be released by treating the resin with steam for a time at 100 o C, and that the resin could be dried and recycled to capture more CO2 with no apparent loss of capacity. However, the energy required to generate the excess of steam needed to release the CO2 in the patent example was significant. This approach would not be sensible for DAC if the energy source to generate the excess of steam required were natural gas (or marine diesel fuel), because the CO2 emissions associated with burning the fuel in this patent example are more than ten times the CO2 captured from the air.
"resin" was mentioned again on pg 57:
One proposed DAC approach is to use a polymer-based ion exchange resin in the OH- form to capture CO2 from the air. The OH- will be solvated by the water molecules in the micropores. An “as received” polymer ion exchange resin has 30-50% water content by weight. In air, the resin adsorbs or loses water, depending on the absolute humidity of the air. The OH- on the solid support is associated with a quaternary ammonium cation bound to the resin, and will be solvated by surrounding water molecules. The observed rate of CO2 uptake is slowed by increased water presence in the micropores [55].
Among the step 4 questions/challenges are:
• Can CO2 sorbents be identified that reduce the CO2 binding strength and/or reduce the increase in order (reduce entropy loss) but remain effective at air capture, and thereby reduce the energy requirement for regeneration and CO2 release?
• Can solid CO2 sorbents be developed that have a greater number of accessible binding sites per unit volume and/or weight?
• Can high-surface-area solid CO2 sorbents be developed with increased effectiveness in the presence of water? Might this be accomplished by minimizing water condensation in the pores to reduce the binding-site blocking effect of water? Or, alternatively, by taking advantage of the high water concentration to increase the effectiveness of CO2 binding or absorb the heat released from the binding reaction?
• Can CO2 sorbents be developed with reduced reactivity with NOx and SOx relative to CO2 for placing of DAC in populated areas?
Well at least the Society of 'Dampening Expectations' had the common decency (on occasion) to provide a list of the technical obstacles and roadblocks
that must be solved, if carbon capture (on a global scale) is ever to become "viable."
But hey, that's what 'hard-headed innovators' do: They go around the obstacles. They remove the roadblocks. They SOLVE the problems.
Assuming they have the means and the opportunity to actually pursue the "Science" that is.
Assuming the mission statements in our Constitution are actually followed:
-- The Promoting Science and the Arts
-- The Encouraging of Inventors and New Discoveries
It's good to have a "mission." Especially one or two, that are
actually Constitutionally-based --
Imagine that!
That would really be a breath of "fresh air" for a change. Actually promoting Science.


