Here is a comparison of the two COVID-19 vaccines developed by Pfizer/BioNTech and Moderna.
General Information and information for recipients
|
Pfizer/BioNTech |
Moderna |
| Vaccine technology |
mRNA |
mRNA |
| Target age group |
16+ |
18+
Testing in progress for 12-17 year olds
|
| Safety for those who are pregnant or lactating |
Not known yet
Animal studies show no harmful effects
|
Not known yet
Interim data from animal studies show no harmful effects
|
| Reduces risk of severe Covid disease? |
Yes |
Yes |
| Prevents asymptomatic infection? |
Not known yet |
Not known yet
Preliminary data suggest yes.
|
| Can vaccinated people transmit the virus? |
Not known yet |
Not known yet
Being studied
|
| Number of doses |
2 |
2 |
| Days between doses |
21 |
28 |
| Side effects |
Injection site pain, redness, swelling and lymphadenopathy; headache, fatigue, malaise, muscle pain, joint pain, nausea, chills and fever.
Higher among younger recipients with stronger immune systems
|
Injection site pain, erythema, swelling, and lymphadenopathy; headache, fatigue, myalgia, arthralgia, nausea/vomiting, chills and fever.
|
| Allergic reactions |
Possible. Check with your doctor. |
Possible. Check with your doctor. |
| Immunity duration |
At least few months
Will take time to determine longer duration
|
At least few months
Will take time to determine longer duration
|
Technical Information and information for healthcare professionals
|
Pfizer/BioNTech |
Moderna |
| Vaccine technology |
mRNA |
mRNA |
| Status |
FDA EUA, also in UK |
FDA EUA |
| Efficacy (point value) |
95% |
94.1% |
| Dose size |
0.3 mL |
0.6 mL |
| Vaccine amount per dose |
30 micrograms |
100 micrograms |
| Doses per vial |
5 |
10 |
| Vaccine preparation |
Thawing; dilution with 1.8 mL of 0.9% Sodium Chloride injection into vial.
Stirred, not shaken!
|
Thawing; no dilution
Swirled, not shaken!
|
| Storage/shipping |
-94oF, -70oC |
-4oF, -20oC |
| Durability after thawing |
5 days at fridge temperature, 6 hours after first use |
30 days at fridge temperature, 12 hours at room temperature, 6 hours after first use |
| Vaccine ingredients |
See below
No preservatives
|
See below
No preservatives
|
| Minimum order |
975 doses
Small rural hospitals may need fewer doses, esp. in the first phase
|
100 doses |
| Approximate cost per dose |
$19.50 * |
$25 - $37 www.biospace.com/... |
| US Govt Funding |
$1.95 billion for 100 million doses |
$4.1 billion for 200 million doses. www.hhs.gov/... |
* Note that in the U.S., people will pay very little out-of-pocket—if anything at all.
Much of the info in the tables is derived from this article by Helen Branswell, although the information is available from multiple sources.
 Images of the Moderna packaging and the Pfizer/BioNTech vial
Images of the Moderna packaging and the Pfizer/BioNTech vial
Vaccine Clinical Trial Results
Here are some more details on the Moderna vaccine trial results from www.fda.gov/…. There is more nuance to the 94.1% overall efficacy number. In particular, the table shows 95% confidence intervals and efficacy across demographic groups. The 95% confidence interval for those over 65 is wider because of the smaller number of participants in that age group. The total number of volunteers is 28,207, evenly split between the vaccination and placebo groups.
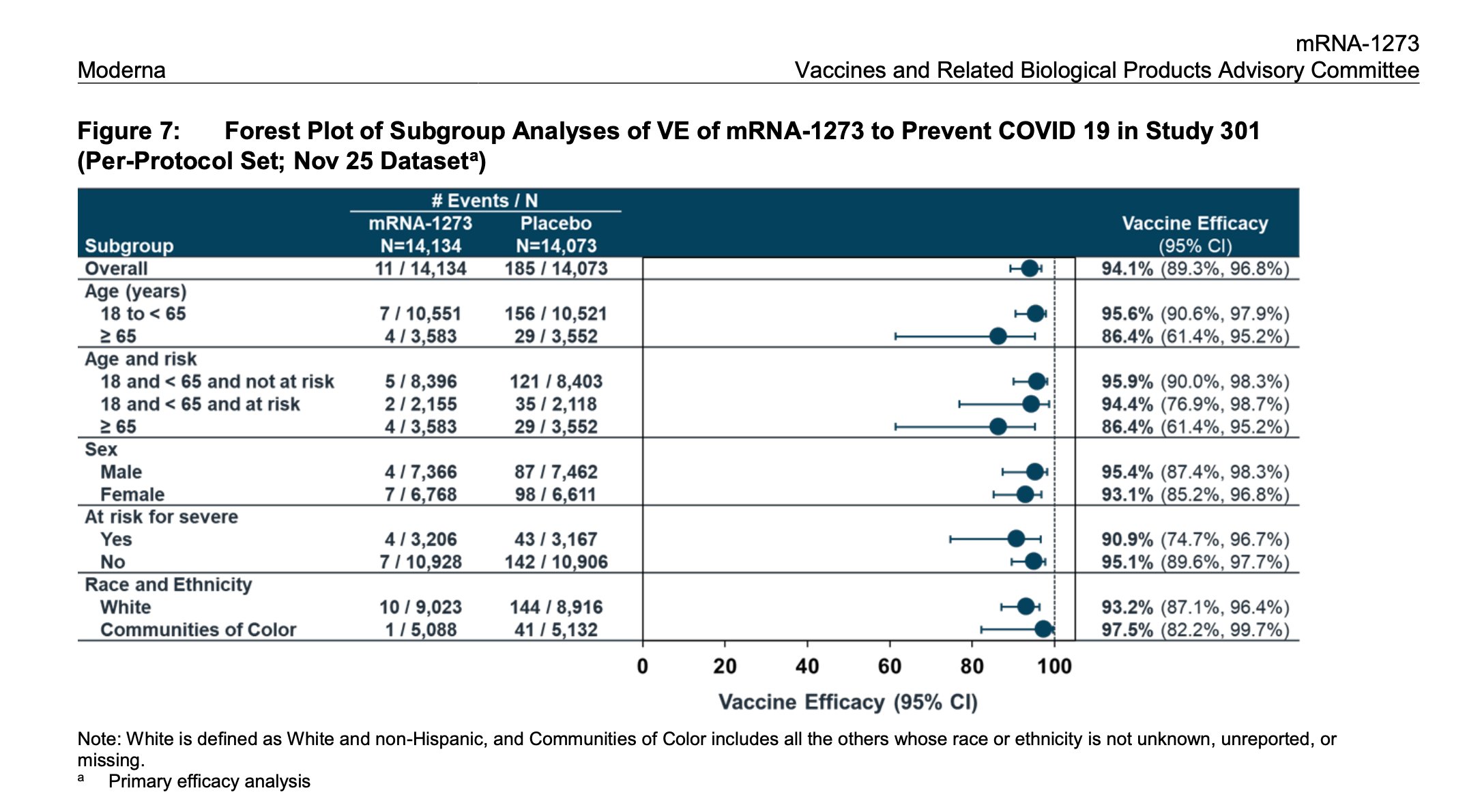 www.fda.gov/...
www.fda.gov/...
The primary efficacy endpoint was symptomatic COVID-19 disease, which was defined as the following:
- At least two of the following systemic symptoms: fever ≥ 38.0°C/100.4°F, chills, myalgia, headache, sore throat, new olfactory or taste disorder; OR
- At least one of the following respiratory signs/symptoms: cough, shortness of breath or difficulty breathing; OR clinical or radiographical evidence of pneumonia; AND
- Documentation of at least one NP swab, nasal swab, or salivary sample (or respiratory sample, if hospitalized) positive for SARS-CoV-2 virus by RT-PCR.
Here is similar data for the Pfizer/BioNTech vaccine from www.cvdvaccine-us.com/.... The 95% confidence interval for those over 65 is wider here as well due to the smaller number of participants in that age group. The total number of volunteers is 37,267, evenly split between the vaccination and placebo groups.
Confirmed cases were determined by RT-PCR and at least 1 symptom consistent with COVID-19 (symptoms included: fever; new or increased cough; new or increased shortness of breath; chills; new or increased muscle pain; new loss of taste or smell; sore throat; diarrhea; vomiting).
Vaccine Ingredients
Pfizer-BioNTech:
- mRNA
- lipids —
- ((4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate)
- 2[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide
- 1,2-Distearoyl-sn-glycero-3-phosphocholine
- cholesterol
- potassium chloride
- monobasic potassiumphosphate
- sodium chloride
- dibasic sodium phosphate dihydrate
- sucrose.
Moderna:
- mRNA
- lipids -
- SM-102
- 1,2-dimyristoyl-rac-glycero3-methoxypolyethylene glycol-2000 [PEG2000-DMG]
- cholesterol
- 1,2-distearoyl-snglycero-3-phosphocholine [DSPC]
- tromethamine
- tromethamine hydrochloride
- acetic acid
- sodium acetate
- sucrose
The lipids are fats, which encase the mRNA fragments and facilitate delivery of the mRNA to cells. The other ingredients works to maintain the pH or stability of the vaccine. www.prevention.com/…
There are no preservatives in either vaccine.
Why the deep freeze?
Both vaccines use mRNA, which is fragile and which deteriorates quickly with temperature and light. Encasing them in lipids and keeping them frozen helps stabilize the mRNA until it is ready for use.
Why the different temperature requirements? It is difficult to tell since the exact formulations of the vaccine are closely guarded secrets.
According to www.npr.org/…, Moderna spokesperson Colleen Hussey explained to NPR in an email that its vaccine doesn't need to be kept so cold because of its particular "lipid nanoparticle properties and structure," and because the company has learned from experience — it's developed ten mRNA vaccine candidates already. "Now we don't need [ultra-cold conditions] as the quality of product has improved and [it] doesn't need to be highly frozen to avoid mRNA degradation," Hussey explained.
It's possible that Pfizer's vaccine could eventually be shown to be stable in somewhat warmer conditions — or for longer times out of the freezer. These ongoing studies will take time to produce results.
5 doses or 6 doses per vial, is that a question?
There has been recent news about how the the Pfizer/BioNTech vial contains enough vaccine for 6 doses and caretakers are throwing away the 6th dose due to lack of guidance. The Pfizer/BioNTech vial contains enough vaccine for 5 doses, 0.3 mL each. But as we can see from the table above, after dilution, the vial contains more than 1.8 mL of liquid. So, technically there is 6 (or even 7) doses in the vial. But there is always some extra amount in vials to account for extra draw in the syringe and wastage. Extracting 6 doses will depend on the dexterity of the nurses to draw precise amounts and avoid wastage. The leftover amount may be less than the required 0.3 mL dose. So, more accurate guidance is needed on how to use the leftover amount.
Can the two doses use different vaccines?
There is no test data for use of different vaccines in the two doses. Healthcare providers with access to multiple vaccines will probably stick with the same vaccine for a given patient.
Who will get it when?
There is a schedule developed by the CDC which prioritizes who gets the vaccine across multiple phases on the vaccine rollout. Phase 1a recommendations were made couple of weeks ago. The group is meeting today to discuss allocations for phase 1b and 1c. Check out the link below for more info.
Epilogue
Which vaccine should you prefer? Most experts state that there is no reason to pick one over the other. Also, most likely, we will not have an option to select the vaccine, especially in the initial rollout. We will get whatever is available with the local healthcare provider.
I hope you find this info useful. Please note that I am not a healthcare professional and I may have missed some of the subtleties in the descriptions of the vaccines and their properties. Healthcare professionals here can add their insights in the comments section.
As always, consult with your doctor and take information from social and popular media with a grain of NaCl.
Further Reading
- A side-by-side comparison of the Pfizer/BioNTech and Moderna vaccines — www.statnews.com/…
- Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Recipients and Caregivers
- — www.fda.gov/…
- Fact-sheet-for-hcp-administering-vaccine-vaccination-providers-full-eua-prescribing-information.pdf — www.cvdvaccine-us.com/...
- FDA Briefing Document Moderna COVID-19 Vaccine — www.fda.gov/…
- Moderna COVID-19 Vaccine EUA Fact Sheet for Healthcare Providers — www.fda.gov/…
- Why Does Pfizer's COVID-19 Vaccine Need To Be Kept Colder Than Antarctica? — www.npr.org/…
- CDC advisory board Recommendations on who gets the Vaccines first — www.dailykos.com/...


