Moderna today announced the preliminary results of a study that shows that its COVID-19 vaccine “retains neutralizing activity against emerging variants first identified in the U.K. and the Republic of South Africa.”
Short summary —
- The vaccine is effective against the B.1.1.7 variant (first identified in the U.K.)
- The study showed no significant impact on neutralizing titers against the B.1.1.7 variant relative to prior variants.
- The vaccine is effective against the B.1.351 variant (first identified in the Republic of South Africa), even though there is a six-fold reduction in neutralizing titers.
- A six-fold reduction in neutralizing titers was observed with the B.1.351 variant relative to prior variants. Despite this reduction, neutralizing titer levels with B.1.351 remain above levels that are expected to be protective.
- The two-dose regimen of the Moderna COVID-19 Vaccine at the 100 µg dose is expected to be protective against emerging strains detected to date.
- The results are based on in-vitro lab testing, not clinical trials.
Moderna’s plan to address new variants -
- 3rd dose: The Company will test an additional booster dose (i.e, a 3rd dose) of its COVID-19 Vaccine (mRNA-1273) to study the ability to further increase neutralizing titers against emerging strains beyond the existing primary vaccination series.
- Booster dose with vaccine against B.1.351 variant: The Company is advancing an emerging variant booster candidate (mRNA-1273.351) against the B.1.351 variant. The Company is advancing mRNA-1273.351 into preclinical studies and a Phase 1 study in the U.S. to evaluate the immunological benefit of boosting with strain-specific spike proteins.
Coverage at the NYT and WaPo —
What does “six-fold reduction in neutralizing titers” mean?
It does not mean that the vaccine is 6 times less effective. Antivirus neutralization titer is determined by calculating the highest dilution of serum that prevents infection. The serum from the vaccinated person is diluted in multiple steps to determine the highest dilution level which is still effective against a certain amount of virus in lab tests. Against the B.1.351 variant, that level was 6x lower than the dilution level that is effective with the current virus strain D614G (i.e., a 6x higher serum concentration was needed). But the level of antibodies present in the original serum far exceeds that in the highest dilution level needed. So, the vaccine is effective. The vaccine will probably lose its potency for B.1.351 a bit earlier, as antibody levels fall with time; that is the main area of concern.
The decrease in neutralizing ability is not the only criteria to assess vaccine effectiveness. There are also T cells, memory B cells and non-neutralizing antibodies, which help protect against the virus and which could not be evaluated in this study.
Info from Moderna about the virus variants
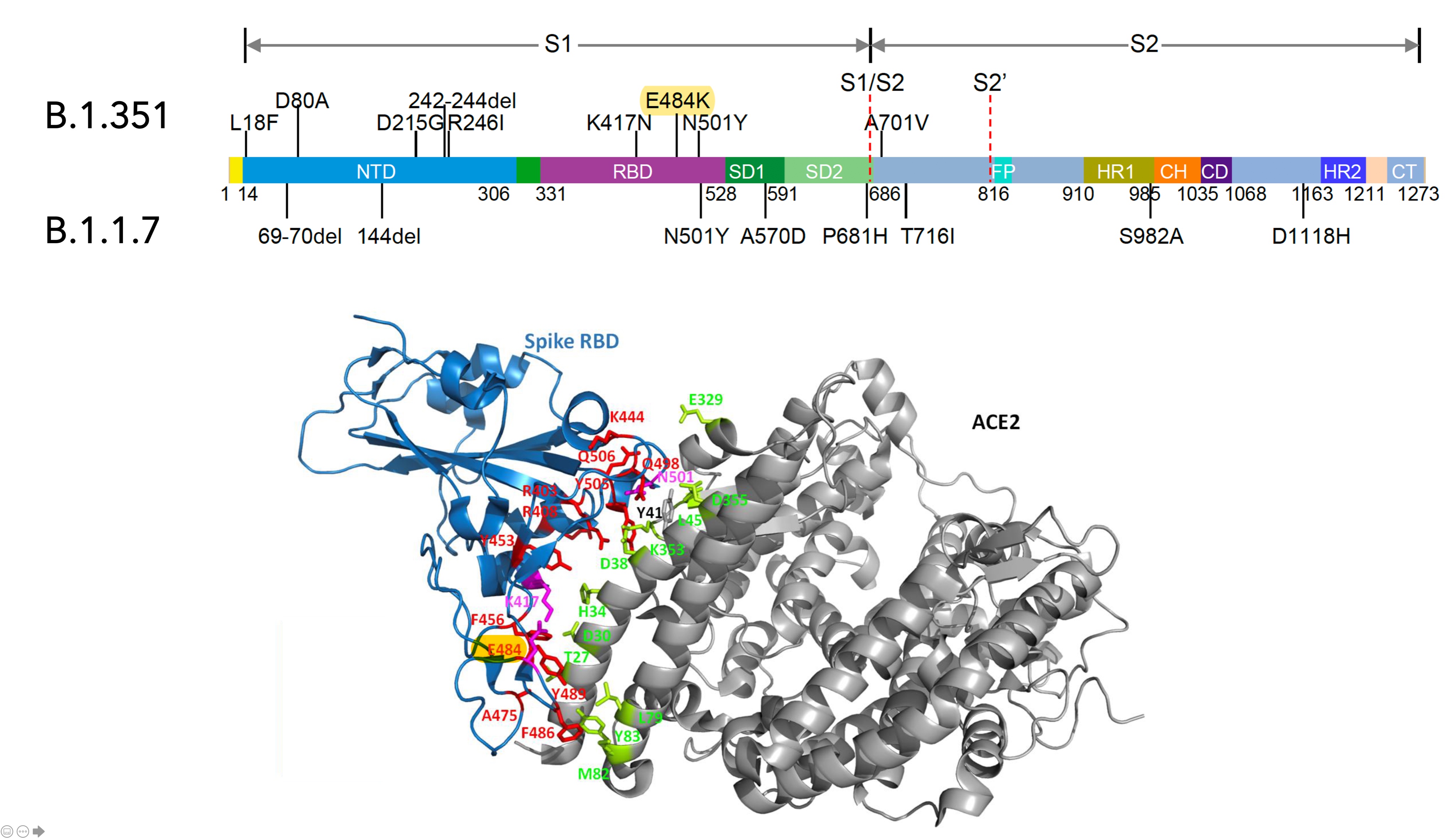
- The SARS-CoV-2 B.1.1.7 variant, first detected in September 2020 in the United Kingdom, has 17 mutations in the viral genome with 8 mutations located in the spike (S) protein.
- The B.1.351 variant, first detected in South Africa, has 10 mutations located in the spike (S) protein. This variant shares many mutations with a concerning variant detected in Brazil.
- Both variants have spread at a rapid rate and are associated with increased transmission and a higher viral burden after infection.
Besides B.1.1.7 and B.1.351, there is the P.1 variant, first identified in Brazil, which has similarities with B.1.351.
A map of the various parts of the spike protein gene and the locations of the mutations.
 Source
Source
Study Details
The study was done using in vitro analysis using blood sera from 8 vaccine clinical trial participants and non-human primates. Sera was collected one week after the second dose was given.
Testing was done using a pseudo-virus, engineered to contain the various mutations of interest. The authors state that “it will be important to substantiate these results by testing the B.1.351 variant in a live virus neutralization assay.”
The study report has not yet been peer-reviewed.
From investors.modernatx.com/… — The in vitro study assessed the ability of mRNA-1273 to elicit potently neutralizing antibodies against the new SARS-CoV-2 variants, using sera from 8 Phase 1 clinical trial participants (aged 18-55 years) who received two 100 µg doses of mRNA-1273, and separately using sera from non-human primates (NHPs) immunized with two doses of 30 µg or 100 µg of mRNA-1273.
For the B.1.1.7 variant, neutralizing antibody titers remained high and were generally consistent with neutralizing titers relative to prior variants. No significant impact on neutralization was observed from either the full set of mutations found in the B.1.1.7 variant or from specific key mutations of concern.
For the B.1.351 variant, vaccination with the Moderna COVID-19 Vaccine produces neutralizing antibody titers that remain above the neutralizing titers that were shown to protect NHPs against wildtype viral challenge. While the Company expects these levels of neutralizing antibodies to be protective, pseudovirus neutralizing antibody titers were approximately 6-fold lower relative to prior variants. These lower titers may suggest a potential risk of earlier waning of immunity to the new B.1.351 strains.
Pfizer/BioNTech
Pfizer-BioNTech released similar data last week (not peer-reviewed), showing that antibody-rich blood serum samples from 16 vaccinated people showed that vaccine was effective at blocking the B.1.1.7 variant. That study did not address the B.1.351 variant.
Like Moderna, BioNTech is also working on an adjusted vaccine, that targets the B.1.351 variant.
Good news from the field
The Pfizer/BioNTech vaccine is providing very positive results in Israel, even against the B.1.1.7 variant.
Bad News from Merck
Merck’s viral-vector based vaccines both fail and will likely be abandoned. Vaccines are hard. Merck has a lot of experience with vaccines, much more than Moderna and BioNTech. Also shows that vaccine companies are honest and diligent.
Epilogue
This is indeed good news and not unexpected. It was expected that the vaccine might be slightly less effective, but not totally ineffective, against the new variants or immunity duration might be shortened.
Yes, the COVID-19 virus will evolve, more so because it is so widespread. It is known to evolve faster in people with weakened immune systems and there is fear that it may evolve faster if the second dose of the two-dose vaccines is delayed. As has been amply pointed out, most variations are not consequential but some are, as we have discovered in recent weeks. But the technology to rapidly create vaccine variants also exists and we are glad that these vaccine companies are acting proactively.
Plus we have competence and a sense of urgency back in the White House.
Of course, we still need to bring down the level of the virus quickly, so that it has fewer chances to mutate into a faster spreading or more lethal variant. Also, the new variants are here and most of the country will not get vaccinated for months. Social distancing and masking are still our best tools to control the virus. Let’s wear a mask, preferably a surgical one or an N95 equivalent. Let’s do it even after we have received the 1st or 2nd dose of the vaccine.


