Two weeks ago, New Scientist created a minor Web phenomenon when they published an article entitled Cheap, safe drug kills most cancers. In the article, New Scientist reported the results of a University of Alberta-led study on the anti-cancer properties of the small molecule dichloroacetate.
The article's contents probably wouldn't have set off the firestorm that it did had it not contained the following two sentences:
The next step is to run clinical trials of DCA in people with cancer. These may have to be funded by charities, universities and governments: pharmaceutical companies are unlikely to pay because they can’t make money on unpatented medicines.
I'll try to explain the basics of cancer biology and why the community's reaction was, in my opinion, a gross over-reaction after the break.
First, some history of the phenomenon on Daily Kos. After the New Scientist article was published, Randular wrote a diary, Potential cheap, safe cure for cancer: Will Big Pharma Allow It? that focused on the Two Sentences from before the break. Randular wrote:
Again, if proven effective, will we ever see it in use in this country? Will patients have to take 'DCA tours' to Canada for treatment?
Within the comments a fervor (and some skepticism) developed that propelled the diary to the rec list, focusing on the meme that Big Bad Pharma would never allow such a drug to take hold in the US. Some anonymous comments (you can follow the link back to the story if you want to find the authors):
I know that cancer is a complicated beast. But if this stuff works even a little bit, wouldn't it be worth a few million in research dollars? I'll wager those dollars will be spent... but in Canada or Sweden or Japan or somewhere that patients lives are actually valued.
The NIH is now nothing but a tool of Big Pharmas. They use taxpayer dollars to develop the meds the companies want, and then make sure only those companies have the right to bid for production.
I presume that DCA is an approved therapeutic agent for the metabolic uses; there isn't anything that would prevent a doctor from prescribing it at such--although I suppose there are some insurance plans that wouldn't cover it.
There has already been debate on the issues raised in the comments and in subsequent diaries, such as Hobbitfoot's excellent diary Cancer cures and the evil of false hope.
Before I give my input into these issues, let me give you some background information on myself. I am a molecular biology graduate student (Ph.D.) in my first year. I do not study cancer, but like all molecular biologists I am familiar with the basics. So I am not an ultimate authority, but I do think I can provide context and a more informed point of view than your average Joe.
That said, let's get into the meat.
Cancer is largely a modern disease. While juvenile cancers do occur (cancer is a top-3 killer until age 15), cancer really starts to wreak havoc as people get older -- older, in fact, than our historical life expectancy. This is significant. It means that the selective pressure for anti-cancerous genes has been very light until recently. In plain English, this means that anti-cancer genes didn't confer much of an advantage to our ancestors because they died too young to use them, decreasing the frequency of these genes and resulting in the rather high susceptibility to cancer that we have today.
(As a side note: when one says "anti-cancer gene" in this context, it doesn't usually mean a new gene that a previous generation didn't have. It mens a different version of a previously existing gene that tilts our cells away from cancer, such as a more active form of a DNA damage-fixing protein.)
Why is cancer a disease mainly of the elderly? The reason for this gets to the root of what cancer is in the first place -- a loss of the normal control of proliferation of our cells. Step back for a moment and compare yourself to a bacterium. The goal of the bacterium is simply to divide as fast as possible and maintain as large a population as possible. We, on the other hand, are multicellular animals. Our cells have a carefully orchestrated pattern of dividing that maintains a relatively constant number of cells throughout our adult life. There is relatively high turnover in the skin and intestines, but low turnover in the brain and heart. But the important new word here is turnover -- cells aren't trying to multiply for the sake of multiplying. They multiply to replace older or damaged cells.
This is a careful balance, mediated by a beautifully intricate signaling and control system. For an example of what a "signaling and control" system means, consider when you cut yourself. The physical process of cutting yourself rips apart cells that were once connected. And, importantly, it rips apart the dense, fibrous material that sits between most of our cells -- the extracellular matrix. When that material is ripped apart, it releases tiny little molecules that tell surrounding cells hey, we just had some trauma! Fix us up! And the surrounding cells are released from their normal restriction against cell proliferation in order to heal the wound.
In addition to pro-proliferative signals, there are also anti-proliferative signals, which can either be intracellular or extracellular in nature. Because this is getting boring pretty quickly, I'll focus on arguably the most studied cancer pathway: the p53 pathway.
p53 is one of those fascinating proteins that has been the subject of an enormous amount of study since it was identified as a tumor-suppressing (anti-cancer) protein (originally it was thought to be oncogenic, but then we realized we had it backwards). The short version of the story is that among p53's roles is its role in responding to DNA damage. Say you're laying out at the beach, baking in the sun without enough sunblock on. While you're warm and happy, the DNA in your skin cells is being steadily damaged (one kind of damage from UV is the formation of abnormal covalent bonds between thymines, the T in A, T, G, and C of DNA). Unbeknownst to you, p53 has gone to work. It has sensed the damage in the DNA and told the cell don't divide! Your DNA's damaged! The result is that you don't get skin cancer (or you do, if the damage overwhelms p53 or a cell happens to have a bad copy of p53).
(Another side note: DNA damage is primarily bad during division. If damage is caught before division, the cell can often repair the DNA to its original state. After the cell divides, any mutations that were caused by the damage are written in stone in one of the daughter cells)
But DNA isn't just damaged from the sun. DNA damage also happens at a slow, steady rate from numerous sources. And this damage builds up over time, because as I mentioned, once a mutation is passed on to a daughter cell, it's there forever. This is why cancer is a disease primarily of the elderly. As mutations accumulate, the chance that a cell will have a mutation that destroys the normal control of proliferation increases, and your chances of developing cancer increases.
Let's return to p53. Its first role in responding to DNA damage is to halt cell division. But if the damage is not repaired fast enough, the cell will actually commit suicide to save you, the host, from death from cancer. This process is called apoptosis, and it is the focus of the New Scientist article.
The real title of the academic article being reported on was A Mitochondria-K+ Channel Axis Is Suppressed in Cancer and Its Normalization Promotes Apoptosis and Inhibits Cancer Growth. To make another long story short, the apoptosis (cell suicide) pathway includes proteins that are embedded in the membrane of the mitochondria. Apparently, when glycolysis overtakes energy production for the cell and the mitochondria turn off, this apoptotic pathway is inhibited. And the study also suggests that dichloroacetate turns the mitochondria back on (by restoring the proper electrical balance of the mitochondrial membrane indirectly), restoring this part of the apoptotic pathway. The result is a cell with damaged DNA that can suddenly kill itself as it is supposed to.
Now that you understand to some degree what the article is talking about, I'll just spit out some thoughts about why I think our reaction was overdone:
Remember that I said p53 is mutated in up to half of all cancers? It is significant that this number isn't 100%. The reason is because I've only mentioned a minuscule number of players in the massive beast that is cell cycle control and the apoptotic pathway. Cancer is the result of multiple mutations acting in concert. Between types of cancer, the genes that are mutated will differ. Between the cells in a single cancerous growth, the genes that are mutated will differ. Our understanding of exactly which genes are mutated and how often is largely limited to research targeting specific proteins of interest (like p53), rather than a more global understanding. In fact, the Broad Institute of Harvard and MIT was recently awarded a $200M grant from the NHGRI (part of the NIH) to sequence cancer strains so that we might have a more global understanding of the roles of different mutations.
Now can you understand why scientists are so skeptical of cancer "cures"? A treatment that attacks p53 (and probably also PDK2, the target of DCA) will only affect a subset of cancers, and might only affect a subset of cells in a single tumor. In a tumor where only some cells are affected the remaining cells will, via natural selection, then dominate the cancer and make subsequent targeted treatment less effective. This is why to this day the most common chemotherapies are basically clear-cutting drugs that attack all dividing cells, including the cells in your gut and your hair. So while dichloroacetate may indeed be a potent anti-cancer treatment, I am personally very skeptical that it will turn out to be a cancer cure. All that is required is one cancer cell to be immune to the effects of the DCA, and the surviving population will proliferate into a DCA-resistant cancer.
Just as important is the fact that no study with DCA has been carried out on humans. While in vitro tests and animal models do provide information on the anti-cancer activity of a drug, it is absolutely not a guarantee that it will behave the same way in a human as it does in a mouse or a tube. Thus, DCA simply is not a human cancer therapy at the moment, and it will not be until these human studies are carried out. This is why the University of Alberta page on DCA says:
At this point, the University of Alberta, the Alberta Cancer Board and Capital Health do not condone or advise the use of dichloroacetate (DCA) in human beings for the treatment of cancer since no human beings have gone through clinical trials using DCA to treat cancer.
In addition, I'd like to vehemently squash the notion right out that the NIH is somehow in the pocket of Big Pharma to the degree that they would never fund a study into the efficacy of DCA. This is simply not true, and to peddle in this falsehood makes us liberals look like tin-foil hat-wearing loons. The NIH pays an enormous sum to study cancer every year, including studies into potential anti-cancer drugs.
As for the patent issue, drugs that have a well-established history can be patented as far as I know, but the patent covers specifically the use of the chemical for a novel purpose (such as cancer treatment). This is why fluoxetine hydrochloride was patented for use against depression (and given the trade name Prozac) and separately patented for use against pre-menstrual dysphoric disorder (and given the trade name Sarafem). Same drug, different name, different patent. So while doctors could probably prescribe DCA off-label for cancer if it pans out (which at this point would be entirely unethical), there is some degree of patent protection that the discoverers could enjoy and could license to Big Pharma. Of course, I am not a patent lawyer, so this information is just my understanding as a scientist.
As for the future, keep holding out hope for DCA, but do not expect it to be a cure for cancer, and don't believe that research on it is being suppressed. If you want to help the scientists studying DCA's efficacy, you can make a donation to the University of Alberta and even specify on the second page that you'd like your money to go to "DCA - Cancer Research Fund".
And also remember that everything I've described in this diary is a scientific model. It is not fact. It is merely the story we have developed that best explains the large amount of data we have accumulated over the years. Some aspects are virtually certain (such as the notion that p53 halts the cell at a cell division checkpoint) while others are higher-level abstractions (such as the idea of a mitochondrion turning "on" and "off" as if from a light switch). So approach everything with an open, skeptical mind as all good scientists do.
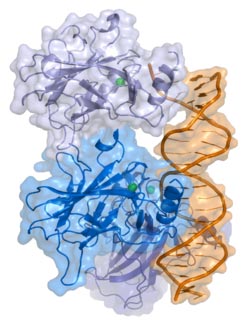
p53 (blue) in complex with DNA (orange) (picture licensed under Creative Commons Attribution ShareAlike 2.5 and graciously taken from Thomas Splettstoesser here).


