My last diary concerned the effects of stress hormones on the reward system, how that relates to the "rush of war," the difficulties of readjusting to civilian life, and the propensity of trauma victims to seek escape in drugs of abuse. This diary continues in that vein, looking at how stress steroids not only amplify the reward system while they are on board, but how they participate in long-term plastic changes in that system.
The Reward System
In the last diary on the rewarding aspects of stress, I showed how elevations in stress hormones amplify the output of the brain's reward system, meso-limbic/meso-prefrontal dopamine. Here is that neuronal system originating in the ventral tegmental area (VTA) and projecting to the nucleus accumbens and the prefrontal cortex.
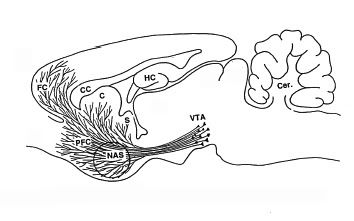
The Steroid Rush
Let's take another look at the effects of elevating stress hormones on dopamine efflux from this reward system. In the panels below (redrawn from Barrot et al, 2000, rats were either intact, had their adrenals removed to prevent endogenous synthesis of the stress steroid, or the adrenals were removed and the animals were replaced with stress levels of the steroid. After taking baseline readings of dopamine efflux in the nucleus accumbens (shell region), the arrow indicates when the animals were injected with either morphine or cocaine.
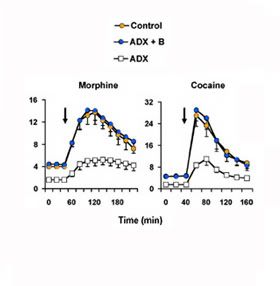
The intact rats are in orange, the ADX + steroid rats are in blue, and the adrenalectomized are in white. First, notice that the drug injection increases dopamine efflux in all rats. However, notice how severely blunted efflux is in the rats without the ability to produce a stress response, whereas in rats capable of secreting steroid or rats replaced with stress levels of steroid, dopamine efflux is substantial and long-lasting. That is what drug users call a "rush." And you don't get the rush without the steroid on board.
However exactly steroids promote dopamine efflux, it seems to be a fast, non-genomic, membrane-mediated effect.
Sensitization
If stress steroids simply made for better rushes, that would be one thing; but they are also involved in issues of long-term behavioral and neuronal plasticity. Take a look at these neurons from the core and shell of the nucleus accumbens after rats have been given a series of saline (S) or amphetamine (A) injections:
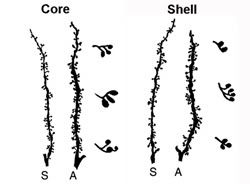
These are dendrites ("antennae" or the receiving end of a neuron) from medium spiny neurons. Note how the spines on the dendrites of the rats receiving amphetamine have become utterly hypertrophic, like Arnold Schwarzenegger as a six-time Mr. Universe (also on steroids, but his were probably not stress steroids but sex steroids; still, steroids are bad-ass organizers--maybe we should get them involved in an impeachment movement!). These neurons are hyperplastically blown-out. Oh, and those little "bushes" to the right of each hyperplastic dendrite are magnifications of individual spines, which have themselves bifurcated into multiple branches. The same thing happens in prefrontal cortex. That is a lot of added potential for receiving drug-related signals, such as those predicting further drug opportunities. This neuronal sensitization accompanies behavioral sensitization to psychostimulants. All the evidence I know points to steroids in behavioral sensitization to drugs (some will argue with this), but as yet there is no evidence I know that directly implicates stress steroids in this particular hyperplastic effects on neurons. Or is there?
Stress Steroids and CRF on Neuronal Plasticity.
While there is no evidence of stress steroids being involved in hyperplastic neuronal changes in this particular form of super-plasticity (more to come in other systems, later diaries), there is definitely evidence that stress molecules play roles in some key types of plasticity in this reward system.
That cartoon above showing dopaminergic projections from VTA to nucleus accumbens and prefrontal cortex does NOT show the projections going back to VTA. The prefrontal cortex sends excitatory projections back to VTA to pump up the volume and cause plasticity within VTA. This is basic fast neuronal transmission. While stress steroids by themselves do not affect firing of VTA dopaminergic neurons, look at what they do in the presence of excitatory juices from prefrontal cortex.
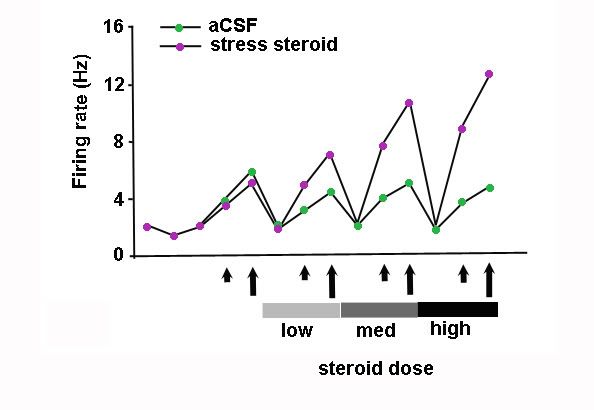
This figure (redrawn, Cho & Little, 1999) shows the firing rate of VTA neurons. Along the horizontal axis you'll see short and long arrows, which represent the application of excitatory juices, a low or high dose, respectively, to a slice of VTA sitting in a dish of artificial cerebrospinal fluid (aCSF). Notice first how firing rates don't change without the excitatory juice. Steroid alone won't do it. When the excitatory juice is added, neurons sitting in plain fluid (green dots) increase firing according to the dose. When the stress steroid (purple dots) is added to the dish in low, med, or high doses, suddenly the neurons really start cracking, then crack some more as the dose increases. This will lead to long-lasting changes in the capacity of these neurons.
Now, you'll remember the diary in which we showed that persistent elevations of steroid can persistently elevate CRF (the anxiety juice) in the brain. A very similar thing happens in VTA when CRF is applied to these VTA neurons in the presence of excitatory fast transmitters:
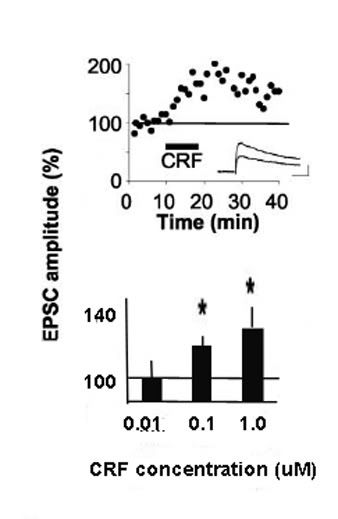
The top figure shows the increase in the amplitude of currents (excitatory post-synaptic currents) within the VTA following the application of CRF. The increased amplitude is rapid and fairly long-lasting. The bottom figure shows that this amplitude increases with increasing doses of CRF.
This is important, because one of the main factors leading to relapse following abstinence is stress. It thus appears that stress-induced drug seeking may involve potentiation of a serial process of CRF glutamate-DA response.
Together, GCs and CRF may act on DA neurons both independently (in parallel) and serially through stress steroid-mediated upregulation of CRF function.
Let's put it all together. Stress heightens the drug rush. Stress causes plasticity within the reward system. Stress potentiates CRF signaling. CRF amplifies VTA firing. Elevations in steroid and/or CRF can create drug cravings.
Reward is Relative
Let me add one final thought. The rewarding value of many incentives is not absolute, but relative to the background context of reward. Thus, supernormal rewards, such as drugs of abuse can make normal, everyday rewards less rewarding by comparison, perhaps in the same way that the rush of war can make everyday civilian life seem sloo-o-o-w, and boring.
By analogy, consider the phenomenon of perceptual contrast below. Here, there are two equal patches of grey within two different contexts. When comparing them, the one on the dark background appears lighter than the one on the light background, even though they are the same.
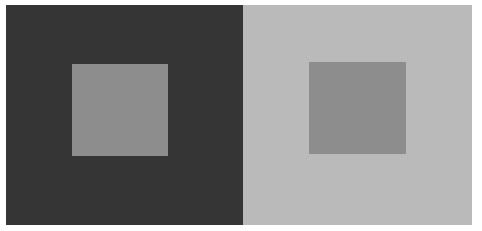
The same type of contrast occurs when the background is a drug or stress-induced memorial state, rather than a perceptual state. And that's why trauma and addiction are a bitch.


