Unless you've been living under a rock, or perhaps in a Secret Undisclosed Location, you know that climate change is probably the great challenge of the century. If we cannot address it successfully, dealing with issues such as poverty, hunger, social justice, etc become impossible.
Ultimately climate is controlled by the energy budget of the Earth's surface. What makes it a complex phenomenon is that a great many things affect that energy budget: landmass layout, orbital tilt, atmospheric composition, ocean currents, reflectivity (albedo) of the Earth, etc. None of these are static: most factors are affected by others on a huge range of timescales that span many orders of magnitude.
Let's look at one such factor, the carbon cycle.
Carbon is particularly important to climate primarily through atmospheric composition. As CO2 (and methane, CH4) it is "radiatively active" -- basically it traps heat in the lower atmosphere, the so called "greenhouse effect". Carbon forms the building blocks of life, which is a sink of atmospheric carbon, by converting that carbon to biomass, some of which gets buried and sequestered from the atmosphere and which results is an atmosphere full of oxygen. As limestone (calcium carbonate) it is a long lived reservoir of atmospheric carbon.
To keep things tractable, we'll need to start with an introduction to ...
Biogeochemical cycles
Already we see that carbon exists in the atmosphere, the Earth, and in living things. It is also present in water. This makes for a complex system, usually described by a box model. A box model consists of a whole bunch of boxes, aka reservoirs. Atmospheric carbon is a reservoir, as are marine organic carbon, surface ocean inorganic carbon, the terrestrial biosphere, etc. These reservoirs are connected by fluxes, flows describing the transfer of stuff throughout the various reservoirs. Reservoirs are usually described by mass, fluxes by mass/time. A flux describing inflow to a reservoir is called a source; a flux describing outflow is a sink. Obviously all sources to a reservoir are sinks of other reservoirs, and all sinks are sources of other reservoirs. Also keep in mind that the magnitude of the fluxes and the size of the reservoir are not correlated. But today we'll just take a look at reservoirs.
(If you are a physicist the term "flux" will make your teeth grate, because as everyone knows a flux is something/(time area), so here the term is really an area-integrated flux. But everyone in the community calls them fluxes, so I will too. Even as my teeth grate.)
The study of the transformation of elements through the various parts of the Earth system is known as biogeochemistry. Biogeochemical models are generally closed wrt mass: stuff will move through the various reservoirs, but the total mass of stuff is conserved. Here is a typical representation of the biogeochemical cycle of carbon (taken from Rohde et al., Earth System Science, J. Wiley and Sons, 2001):
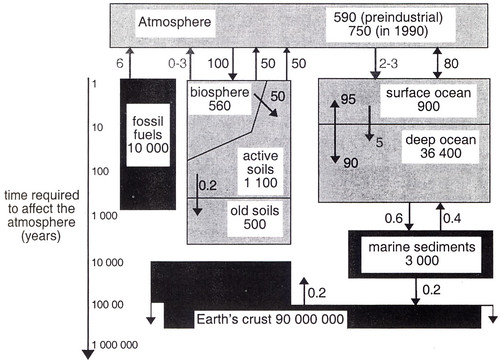
A word about units. There is a lot of carbon in the Earth system, so the typical units are in 1015 grams = 1 petagram = 1 Pg = 1 gigaton = 1 Gton = 1 billion metric tons. Fluxes are usually in Gton/yr.
In the technical literature the mass typically refers ONLY to the mass of carbon: 1 Gton atmospheric carbon = 3.67 Gton CO2. (Carbon has a molecular weight of 12 g/mol, CO2 has a molecular weight = (12+16+16) = 44 g/mol, or 3.67 times more). AFAICT there is no convention in the general literature, so be careful when you see numbers: they may mean total amount of carbon containing stuff, or they may refer to only the carbon content. The numbers in the figure above refer only to the carbon content. Some of the numbers are a bit out of date -- the figure is 15 yrs old (I like it because it explicitly includes the lithospheric reservoir).
Note that this is a closed system with respect to carbon (in the figure above the fossil fuel reservoir is actually a part of the Earth's crust), and that all fluxes entering a reservoir are balanced by fluxes exiting that reservoir. If that is not the case, the reservoir must either grow or shrink. Because the whole system is interconnected, a perturbation (a change to either a flux or reservoir size) will ripple throughout the entire cycle. If the system is in ~equilibrium, even small perturbations can create significant changes in at least parts of the system: how much depends on the relative timescales involved.
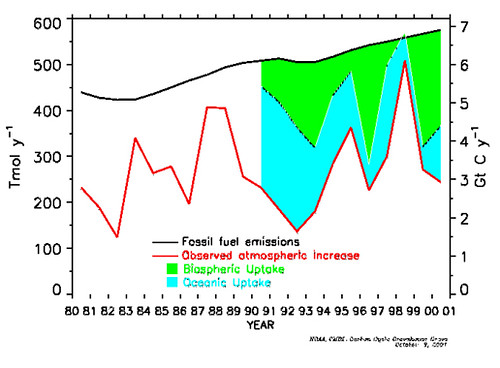
Today the estimate for marine C emissions to the atmosphere is about 90 Gton/yr, whereas fossil fuel combustion in 2005 was about 7.7 Gton/yr (warning Excel file). There are another ~120 Gton/yr C coming out of the terrestrial biosphere and soils. But there is also a marine C uptake term, of about 92 Gton/yr. In preindustrial times the uptake flux was about 90 Gton/yr, to balance the emission -- today the oceans are trying to take up the additional C emission, but can't quite keep up. The natural terrestrial uptake term is a bit more than 120 Gton/yr, balancing the emission and also trying to take up the FF term. Excluding the FF emission term, the large marine and terrestrial emissions are balanced by large marine and terrestrial uptake: that must be true or the atmospheric reservoir would have been growing or shrinking, which we know was not the case from ancient air bubbles trapped in the ice. So while the fossil fuel emission term may be small in gross terms, it (along with a land use term) is entirely responsible for the net emission term. To reduce atmospheric carbon we need to increase sink terms or find a new one. Here's a figure from NOAA estimating how the Earth system is trying to accommodate that net emission:
Reservoirs
There are a good many ways to partition the reservoirs, the simplest is to acknowledge the 4 great domains of the Earth system: the atmosphere, the hydrosphere, the biosphere, and the lithosphere. I'll use the reservoirs shown in the figure above, but keep in mind that that is not the only way to do it, and the best way depends on the question you are addressing. Also keep in mind that the magnitudes of the reservoirs are not as well known as we would like, and there is a fair bit of guessing involved. But current estimates for total carbon are on order 100000 Gtons, nearly all in the lithosphere, and nearly all the rest in the oceans. But we'll see without the tiny quantities in the biosphere, things would be very very different.
Atmosphere
Atmospheric carbon is almost all (99%) in the form of CO2. There are about 384 parts per million CO2 (out of 1 million air molecules 384 are CO2) and climbing; 1.8 ppm methane, or CH4; 0.02 to 1.5 ppm CO (really clean to really dirty air); and everything else -- ethane, propane, acetone, formaldehyde, isoprene, etc -- add up to less than 0.01 ppm. Methane is actually the more efficient GHG per molecule (it has the larger infrared absorption), just how much depends on timescale. On a ~century timescale it's about 25 times more effective. Methane has a residence time of about a decade; CO2 doesn't have a single residence time, but components spanning weeks to tens of thousands of years. More on timescales later.
The mass of the atmosphere is about 5.15 x1018 kg. The molecular weight of air is about 28.97 g/mol, so that's 1.78 x1020 moles of air. Out of every million moles, 384 are CO2, or 6.83 x1016 moles CO2, or 6.89 x1016 moles carbon, if we take CO2 to be 99% of atmospheric carbon. Carbon has a molecular weight of 12 g/mol, so the magnitude of the atmospheric carbon reservoir is 8.27 x1017 g or 827 Gtons of carbon (not CO2!). That's actually a slight overestimate, since stratospheric CO2 is a little lower -- 7 ppm or so -- than tropospheric (rate of mixing can't keep up with rate of emission) but keep in mind that the troposphere accounts for over 80% of the atmosphere by mass. Even so, the 750 Gton estimate for 1990 is already woefully out of date today.
Biosphere
The biosphere is comprised of living things. From a chemical point of view, it is the biosphere that controls the oxidation and reduction of carbon in the Earth system. Biological producers take oxidized carbon in the form of CO2 and convert that to biomass via photosynthesis (also much less importantly by chemosynthesis); life then takes that biomass and oxidizes that back to CO2, using the free energy of reaction to power growth, maintenance, and reproduction. (It's not a perpetual motion scheme because our system is not closed to energy: solar energy is an external input, and the entropic tax is incurred outside the system.)
The key reaction in the carbon cycle (and the one that makes Earth unique in the solar system) is
CO2 + H2O + energy <---> "CH2O" + O2
Here "CH2O" is shorthand for carbohydrate (such as glucose, C6H12O6); it's not formaldehyde. The forward direction is photosynthesis, the reverse is respiration. This cycle of oxidation (respiration) and reduction (production) is key to the carbon cycle. Life uses solar energy to create and maintain a chemical potential by providing both reduced material and a strongly oxidizing environment. Without life, carbon would be primarily in the oxidized reservoirs, just as it is on our sister planets Venus and Mars: that is, in CO2 and carbonate rock. The atmosphere would contain trace quantities of oxygen, through the photolysis of CO2 and water vapor. Any reduced carbon buried in the rocks would eventually be uplifted and weathered into oxidized carbon. Life changed everything.
We should also distinguish gross primary productivity (GPP -- the rate of conversion of CO2 to biomass) from net primary productivity (NPP -- GPP minus respiration of primary producers.) NPP is what we want if we are interested in the accumulation of biomass. Producers are also known as "autotrophs"; those that can only do the reverse reaction are freeloaders, spongers, or "heterotrophs". There are in fact other forms of auto- and heterotrophy, based on sulfur, nitrogen, even metals; those will have to await another post.
The figure above does not show the marine biosphere, about 3 Gtons (this is a difficult figure to estimate). It is much smaller than the terrestrial biosphere because there is no need to invest in a massive support structure (ie, wood) when you're living in the ocean. This is reflected in the average composition of marine biomass (the so-called Redfield ratio): 106 parts carbon, 16 parts nitrogen, 1 part phosphorus (also 110 parts oxygen and 263 hydrogen, and trace minerals). It is probably no coincidence that ocean water typically has a N:P ratio of 16:1 -- that is, those critical nutrients are used as efficiently as possible. (There are various numbers for the terrestrial version of the Redfield ratio (C:N:P), all about 900-1000:9:1. Note the much greater need for structural material on land than in water.) Small though this marine reservoir may be, it is nonetheless responsible for the production of reduced carbon in the ocean, transport of that reduced carbon to the deep ocean, and the long term (millions of years) sink of carbon out of the atmosphere through limestone formation and deep-ocean deposition of organic carbon fixed from atmospheric CO2. Here are two examples of primary production (shallow water, in this case): one is turtle grass, the other the phytoplankton that give the green color cast:

Consider the oyster. More specifically, Ostrea edulis and Crassostrea gigas, those succulent briny comestibles so delicious raw with a dash of pepper or cooked into a chowder or deep fried. Ecologically they are invaluable for filtering the water and keeping algae in check and for providing reefs for crabs, fishes, annelids, snails, other invertebrates, etc. They sink atmospheric carbon in 2 ways, indirectly through the consumption of algae, and directly through shell formation. Shells of course are calcium carbonate in the form of aragonite. The first way is quite inefficient, as predation and decomposition of biomass are far more likely than deep burial. The second way OTOH ... the shelly bits deposit and will ultimately turn into limestone. The sink of limestone is uplift and weathering -- this releases carbonate back into the water column where it may taken up by primary producers or by molluscs like oysters. But limestone will only weather in acidic environments, which means either by rainfall or in fresh water (sea water is slightly alkaline), which means you need to wait for geological uplift or a decrease in sea level. Both of those are slow processes, so limestone acts as a long lived carbon reservoir.
Unfortunately the oyster fishery is suffering these days, reeling from a two-pronged attack by environmental degradation and rising temperatures. Today, global catches are down to 4.5 million tons (this is not, of course, the entire oyster population). In some places such as the Chesapeake, catches are down 100 fold since 100 years ago. Let's run some numbers:
About 90% of the oyster weight is shell. Calcium carbonate has a molecular weight of 100 g/mol, of which carbon makes up 12. Because this carbon is precipitated from dissolved CO2 taken up from the atmosphere, discarding the oyster shell to form reefs for oyster spat effectively sinks atmospheric carbon. 4.5 Mtons oyster = 4.05 x1012 g CaCO3 = 4.05 x1010 mol CaCO3 = 4.86 x1011 g C = 486000 tons C. Not much, not by itself going to solve the climate problem, but a silver BB all the same. If we can increase demand for oysters, that would put pressure on estuarine communities to clean up, and even a 10-fold increase takes us up to 4.86 Mtons. So eat your oysters! Environmentally beneficial, climatically responsible, healthy and delicious -- what more could you ask?
Terrestrial biomass is dominated by wood, which is made up of cellulose, hemicellulose, and lignin, all of it made from air, water, and light by the magic we call photosynthesis. Lignin is particularly important because after all these millions of years it remains almost entirely indigestible (the exceptions to that can't live in an aerobic environment so have to find refuge in places like termite guts). Accordingly it serves as a medium term (~200 yr) carbon reservoir. There are about 2-300 times more carbon in terrestrial biomass as there is in marine biomass and some 2.5-3 times as much carbon in terrestrial biomass and soils as there is in the atmosphere. Estimates are tough, especially when we consider root mass -- that is probably underestimated.

Hydrosphere
The hydrosphere accounts for rivers, lakes, ice, subterranean water, and oceans. Of this, the oceans account for the vast majority, over 97%, so I will limit discussion to that.
CO2 is weakly soluble in water, according to
CO2 + H2O <--> H+ + HCO3-
The result is a weak acid (carbonic acid), and pure water in equilibrium with 384 ppm CO2 has a pH of about 5.5, and even pristine rainwater has been getting increasingly acidic over the last 150 years. Bicarbonate is the most common ion in fresh water. In sea water bicarbonate comprises a small fraction of total ionic strength. Ions such as Cl-, Na+, Ca2+ etc are much more prevalent, and these ions case the oceans to be slightly alkaline, with an average pH of about 8.1. This pH is also decreasing with increasing atmospheric CO2, which reflects ocean acidification.
Ocean acidification is not a good thing, at least at the rate at which it is occurring. If it continues it will adversely affect organisms such as calcifying phytoplankton and corals. We'll see that the ocean is not in equilibrium with the atmosphere wrt CO2, and even if we stopped all CO2 emissions it will take centuries for the ocean to achieve equilibrium, with the new equilibrium being a more acidic ocean than what we have now. To stop this we'd have to suck CO2 out of the atmosphere at a rate much faster than the rate of uptake by the oceans. If we cannot do this, the oceans will continue to acidify.
Please note that the ocean will remain basic, just less basic. The oceans do not have to become acidic (pH < 7) for utter chaos to break out.
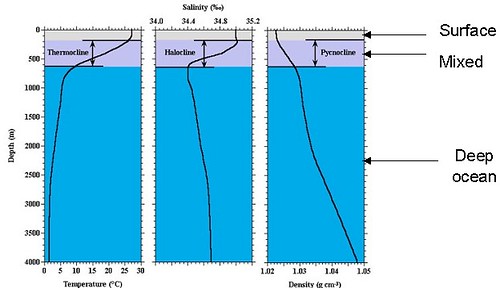
It is useful to partition the ocean into a surface ocean and a deep ocean, because the timescales and chemical processes in each are different. The deep ocean is much the larger reservoir of carbon. There are a number of ways to demarcate the two regimes, depending on the question asked; the one we're interested in is where we have biological production (surface ocean), and where we don't (deep ocean). All production takes place in the surface ocean, as does exchange with the atmosphere. All deep ocean carbon is surface export, either by mixing or settling. Deep ocean carbon is lost to the surface by mixing in upwelling zones, and to the seafloor by settling. The turnover times of the ocean are ~250 (Atlantic, Indian) to 500 (Pacific) yrs. Note that that is longer than the timescale of FF emission (starting ~150 yrs ago and really picked up ~50 yrs ago) so deep ocean carbon is not in equilibrium wrt the atmosphere. Here's a figure to illustrate (I believe originally from NOAA, but I have lost the attribution):
The surface ocean is supersaturated wrt to CaCO3 formation, although nearly all CaCO3 formation is biologically mediated (but see these projections). The deep ocean is undersaturated with respect to carbon, and the deeper you go, the more undersaturated the water. This is why some carbon sequestration schemes involve injection of CO2 into the deep ocean. But keep in mind that the ocean turnover time is on the order of a few centuries, so CO2 injected into the deep ocean will reequilibrate when that water comes back up to the surface. This is a not a long term solution. If you must sequester CO2 in the ocean, at least do it in downwelling zones.
We also need to classify carbon here.
DIC: dissolved inorganic carbon (CO2). CO2 is weakly soluble in water, according to CO2 + H2O <--> HCO3- + H+ <--> CO3= + 2H+. HCO3- is also known as bicarbonate ion, CO3= as carbonate. In both fresh and salt water it is almost all bicarbonate. DIC forms the vast majority of marine carbon, over 97%. All marine primary production goes through DIC, not directly from the atmosphere. This is also what is used to form CaCO3 (shells). CaCO3 is not stable at high pressures and low temps, so below 4-5km depth (the carbonate compensation depth) it dissolves into DIC.
DOC: dissolved organic carbon (aka DOM, for matter). Includes organic acids, aldehydes, lipids, carbohydrates, sterols, etc. There is no comprehensive inventory so apply large uncertainties to reservoir numbers. A typical estimate for total DOM is ~1000 Gtons. DOM has a lifetime of ~1000 yrs, longer than ocean overturning; it is said to be "persistent", largely because of efficient biological recycling in the water column.
POC: particulate organic carbon (aka POM). Particulate organic bits, stuff like dead and decaying plankton, fish poop, slime houses (creatures called larvaceans create houses of mucus, probably to trap food, and routinely discard them when they get cluttered or damaged. These houses are also called "marine snot"). POM can "rain down" the water column, and is sometimes known as "marine snow". While DOM is pretty efficiently scavenged, POM is an important ocean sink of carbon. There is no POM compensation depth so unless "mineralized" (converted to DIC by consumption and subsequent respiration) POM settles to the seafloor. Deep ocean basins near trenches may have 100s if not 1000 m of organic muck from accumulated POM. Subduction of this organic muck is crucial to maintaining atmospheric composition. Again, no good estimates here, typical estimate ~30 Gton.
Planktos recently gave up on an effort to sell carbon mitigation credits by fertilizing the Southern Ocean with iron. The idea was to create a biomass bloom which would then settle as POM to the deep ocean and hopefully seafloor, sinking atmospheric carbon in the process. The effort was abandoned because of criticism that there may be unforeseen side effects. In fact there might be, but even if there weren't they failed to demonstrate the key number, which is carbon export to the deep ocean. Without this there is no way to know how much carbon is actually being sequestered. In fact, biomass tends to be very efficiently recycled, especially in nutrient limited regions (if iron were no longer limiting nitrogen and phosphorus would be) so it is likely that the great majority of the carbon would remain in the surface ocean, which would then reequilibrate with the atmosphere in a matter of weeks. Theirs was a poorly thought out business plan.
Here's some shallow water POM, with the macroscopic calcifier Fasciolaria lilium (the banded tulip snail) hunting among the primary producers Thalassia testitudinum (turtle grass) and unidentified phytopankton over a bed of what is largely the inorganic remains of other calcifiers and organic muck. From the stickiness on leaving the water there was evidently a lot of DOM in it as well. I hope you can see carbon flowing through the various reservoirs: DIC, DOM, POM, marine biota (alive and dead), and CaCO3. (Somewhere it should be mentioned that among the most important calcifiers are actually plankton, like coccolithophores.)

Lithosphere
The lithosphere refers to the Earth's crust and sometimes part of the upper mantle (again, the definitions depend on the question asked). For carbon, the lithosphere is by far the largest reservoir, and the slowest moving, so slow it is nearly decoupled from the rest of the system. Just as is the case in the oceans, there are two types of carbon in the lithosphere: organic (reduced) and inorganic (oxidized). Organic carbon includes shales, tar, kerogen, and petroleum; inorganic is limestone. The source term is primarily biological deposition, generally marine, although wetlands also contribute (coal formation), as do inorganic "whiting" events (nonbiological carbonate deposition). The sink term is through uplift, and can be graceful (weathering of limestone formations such as Mt Everest) or messy (Tambora, Krakatao, Pinatubo ...) Perhaps 80% of lithospheric carbon is inorganic, making that easily the largest reservoir on Earth.
From a climatic perspective both serve to sequester carbon out of the atmosphere in a huge, very slow moving reservoir. But chemically the organic and inorganic forms have different effects wrt atmospheric composition and climate. Let's recall the respiration reaction: CH2O + O2 --> CO2 + H2O. If some CH2O is stuck somewhere O2 can't get to it, such as in the lithosphere, O2 has nowhere to go and builds up. How much O2 there is depends on how much buried organic carbon is available for weathering (the more there is, the lower the O2) It is the burial of organic carbon that provides us with our current 20.9% oxygen in the atmosphere. It is the burial of organic carbon that provides the ozone layer that acts as a UV shield that allowed life to take place on land, that allowed heterotrophy by aerobic respiration. The action of this very large, slow moving, buried reservoir nonetheless has strong repercussions for the atmosphere and biosphere, reservoirs it is very tenuously coupled to. The headbone is connected to the anklebone is connected to the hipbone.
Here's a history of oxygen and CO2 levels in the atmosphere from Robert Berner's GEOCARB model, taken from Peter Ward's Out of Thin Air:
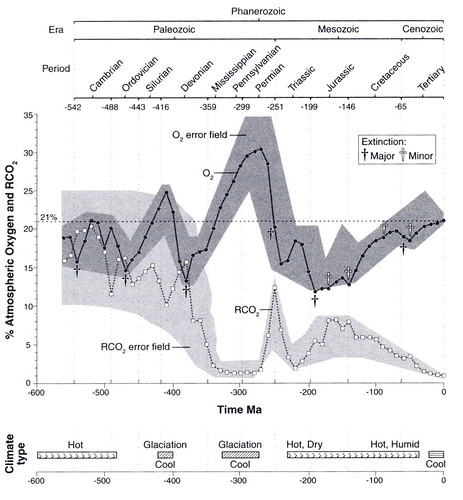
You can see when plants invaded land about 350 MYA from the huge drawdown of atmospheric CO2 and the rise in O2. Most of the great coalfields date to that period (the Carboniferous). It ended with a bang in the Permian extinction. The anticorrelation between O2 and CO2 isn't perfect, but it is pretty good, implying that over the last half-billion years or so the main (but not exclusive) drivers of oxygen levels are the uptake and burial of CO2 and the uplift and weathering of buried carbon.
One last thing. We've noted how the carbon cycle is coupled to the nitrogen and phosphorus and even iron cycles when we were talking about the biosphere. Here's another interconnection, this time with silicon.
CaSiO3 + 3 H2O + 2 CO2 --> Ca(2+) + 2 HCO3- + H4SiO4
Silicate rock weathers in the presence of CO2 and water (carbonic acid) to form bicarbonate and silicic acid. Silicic acid runs off into the oceans where it provides the raw material diatoms use for their shells. The bicarbonate ends up in the water column, sinking atmospheric carbon. If conditions are right the bicarbonate precipitates out as CaCO3, releasing one CO2 back to the atmosphere and sequestering the other.
Ca(2+) + 2 HCO3- --> CaCO3 + H2O + CO2
These reactions are important parts of the long term carbon cycle. There are proposed sequestration schemes that take advantage of these reactions to lithify atmospheric carbon. In principle such schemes would provide into stable, long-term reservoirs, but it is not yet clear whether such schemes are practical.
Fluxes
None of this makes sense without looking at fluxes and timescales. Given the length of what I'd hoped would be a brief overview, however, that discussion will have to await part II.


