Physicists have known about the thermoelectric effect for decades: there are some materials that, when heated, produce electricity. Alas, these materials tend to be both expensive and inefficient. In other words, not much bang for the buck.
But with two interesting new discoveries this week, all that may be about to change. Not soon, perhaps – these are still just a theoretical results in quantum physics, after all – but much, much for the better.
Imagine capturing electricity from the heat of your exhaust pipes, or your brakes, and using it to recharge the battery in your hybrid vehicle. Imagine a solar energy system that converts solar heat directly into electricity cheaper and more efficiently than any photovoltaic cell ever could. Imagine capturing waste heat from a factory, a furnace, or a powerplant, and putting it to good use.
Impossible? Perhaps not for much longer.
A good thermoelectric material allows electricity to flow freely through it, like a metal conductor, but won't allow heat to flow through it much at all (like a thermal insulator). Since most materials are either good at conducting both heat and electricity, or bad at conducting both, good thermoelectric materials are hard to find.
Scientists use a figure-of-merit (called ZT) to describe how good any material might be at thermoelectric generation, which is based on the ratio between the material's electrical conductivity and its thermal conductivity. For normal silicon crystal ZT is about .01, which is horrible. For actual real-world thermoelectrics you can buy off the shelf, ZT = 1 is about the best you can do. For the very best (i.e., exotic) laboratory nano-materials, ZT has been as high as 3. But that's still not good enough; if you really want to change the game for thermoelectric generation, you would need to find something with ZT > 4.
So — how does ZT = 2 sound, using plain old silicon? And if that's not good enough, how does ZT = 50 sound, using quantum effects? And it could go even higher, theoretically.
The first bit of research, scientists Cal Tech use what they call a nanomesh of normal silicon. That means that they take a normal wafer of silicon and drill an array of tiny holes in it. The holes are 11 to 16 nm in diameter and spaced 34 nm apart. This works by slowing down the passage of phonons. A phonon is a packet of vibrating atoms that is the way heat is transferred on the nanoscale. Phonons are measurable and have measurable sizes, and the holes are spaced to interfere with the movement of phonons, while being too small to affect the movement of electrons. That makes the silicon nanomesh much worse at conducting heat, while only slightly affecting its ability to conduct electricity.

Electon micrograph of silicon nanomesh. The black bar is 200 nm long. Visible light has a wavelength of 300 to 700 nm.
Earlier work with silicon nanowires had allowed the Cal Tech team to raise the ZT to 0.6, and the nanomesh approach effectively doubles that – and this is just the first try, so you can expect it to go higher. Since it's easy to make tiny things out of silicon, this could be both cheap, and fast to market.
But getting as high as ZT = 2 from a nanomesh might be a total yawner, if scientists at the University of Arizona are right. Utilizing cool quantum effects, they have shown simple polymer molecules composed of benzene rings that might push ZT up to 50 or even higher.
The trick occurs because of the dual nature of matter at the quantum scale: electrons (like photons, and everything else for that matter) behave both like particles and like waves. And when they behave like waves, their position is uncertain. In fact, an electron can be in two places at once, as long as you're not measuring it to find out exactly where it is.
Benzene is a hexagonal ring of six carbon atoms, with six hydrogen atoms studding the rim. Like most organic molecules, benzene isn't good at conducting either heat or electricity. The Arizona scientists did something a little odd: instead of running the electrical current from one end of the ring to the opposite side, they ran the current between two offset carbon atoms on the ring. This meant that there were two possible paths for the electron to take: either the short way (crossing 2 carbons) or the long way (crossing 4 carbons), around the ring. And since electrons are subject to quantum effects at this scale, a single electron goes both ways around the ring -- which means that the two "parts" of the electron don't interfere with each other when they get to the other side. And that's good, because if the two "parts" had traveled the same distance around the ring, the interference might be destructive interference, and when that happens, you don't have your electron any more. (Ask a quantum physicist to explain why).
So the asymmetry sharply reduces the chances of destructive quantum interference, which sharply increases the electrical conductivity. And that's just the beginning, because benzene forms polymers (that is, long chains) quite easily. These polymers are a form of plastic, and the longer the chain gets, the worse the thermal conductivity gets. But you can arrange it so that every ring of the chain forces one of these asymmetrical choices for the electron, so the electrical conductivity stays high. That means that the longer the polymer chain gets, the higher the value of ZT. For a chain of just 2 benzene rings, ZT is computed at about 2 and thermal efficiency at about 27% (higher than most gasoline engines). For a polymer chain of 30 benzene rings, ZT should theoretically be around 50. (I should emphasize that this is all theoretical at this point; there may very well be practical considerations that make this approach difficult or impossible in practice.) And benzene isn't the only molecule which should have the same property.
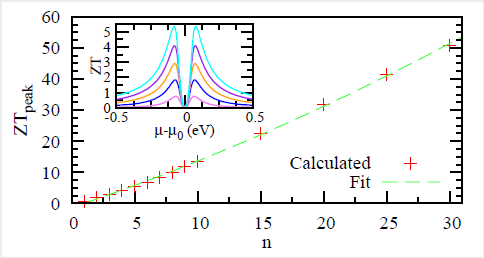
Graph showing how ZT increases with the number of benzene rings in the polymer.
The exciting thing here is that benzene, too, is quite cheap, and the methods for making a polymer out of it have been known for decades. All you would have to do is grow a layer of benzene polymer on a conductive substrate and put another conductive layer on top; the electricity would flow from one to the other, using the heat of the lower layer to drive the process. And the layer is just microns thick, so you could put it anywhere there is a source of waste heat.
Wow!


