Halloween; All Hallow's Eve. Ghosts and witches, creepy spiders and black cats! Lots of science fun in them there legends, some of which I will be exploring the month of October in Science Friday. Mrs. DarkSyde of course doesn't much care about the hard science, but she is one of those who loves fantasy sci-fi: Especially Lord of the Rings type novels. So choice of costume is, needless to say, a topic of great import here at DarkSyde Manor. Meaning I'm stuck with playing a supporting role to whatever strikes her theatrical fancy.
Sadly, Lady DS was struck with one of the worst fevers she's ever had on Hallow's Eve 2004. She never got to show off her threads. So 'we' decided that we'd use the same attire this time. I was told to get the costumes together yesterday, and discovered a problem with mine. A twist most interesting, oddly coincidental, and deliciously scientific, given my assigned Halloween character. I was eager to share all that history and science with my better half. Alas, once again, I was waved off by The Hand of Spousal Boredom. And here I am ...
(
Science Note- This Sunday, watch Duncan Black interview Dr Paul Myers on EvolveTV. Subject: Intelligent Design Creationism)

Cinnabar crystals under low power magnification
Lost in the mists of prehistory, thousands of years ago, a reddish substance became all the rage in Eurasia. The amazing mineral was a source of dye, came in cool crystalline form, and quickly became a source of fascination among natural philosophers. Samples have been found in Egyptian and Chinese tombs dating well before the Christian Era. It was called cinnabar.
The Greek philosopher Empedocles formulated a forerunner of the periodic table in which substances were made out of various combos of fire, air, earth, or water. Seems quaintly inaccurate nowadays. But think of it as a cogent attempt to categorize the fundamental states of matter -- solid, liquid, gas, and plasma: Not a bad try for someone stuck in the fourth century BC! Empedocles' idea persisted until the advent of modern chemistry and is still around today in vestigial form as classifications used in astrology.
Early Mediterranean City-states valued cinnabar as a semiprecious stone and cosmetic pigment, and soon learned to isolate another wondrous enigmatic material from it. The new substance was both a liquid and a metal--water and earth! Early natural philosophers felt it held the secret to transmutation of metals making it the key to wealth beyond dreams, the so called 'philosophers stone'. Today we know it as an element with the chemical symbol Hg reflecting the ancient name, Hydrargryum, meaning watery or 'living' silver. Thus it came to be known in the western world as 'quicksilver'. But hydrargyrum is more commonly called Mercury today.
A shiny metal that flows like water; Elemental mercury is just plain fun to play with! Quicksilver has such a peculiar blend of properties that it played a prominent role in early alchemical experiments of all kinds. And of course some of those early alchemists also ran a lucrative con on the side, in which they would try and convince the rich that they were on the verge of being able to make unlimited quantities of gold from lead or tin. Mercury was usually involved in a sleight of hand scam where small amounts of precious metals would appear after an exhaustive, technologically dazzling presentation: Followed by a plea for investment capital to 'make the process economical and you richer than the Vatican M'Lord!" And off the alchemists would be to fleece the next credulous and greedy Nobleman. In addition mercury was used in beds and other items adorning the chambers of opulent halls from Europe to Persia.
It was the Romans who found that exposure to cinnabar came with a few, shall we say, drawbacks? Laborers in those mines lived on average for two or three years before succumbing to a variety of symptoms including termors, extensive skin rashes, breathing problems, dementia, intense pain similar to Kidney stones and on and on. In the end the pitiful victims were sometimes stark raving crazy, babbling to themselves incoherently and shrieking at paranoid hallucinations while twitching uncontrollably and soiling themselves. Not a pretty sight and not the way anyone would want to go out. A sentence to the cinnabar mines meant lingering death, sometimes even for the unfortunate Roman guards.
Mercury poisoning it seemed could drive a person insane and afflict the poor bastard with a hodgepodge of unpleasant ailments along the way. During the Dark Ages and medieval era perhaps this caveat was forgotten or ignored: Early alchemists used their sense of taste as an analytical tool, ingesting minute bits of all kinds of nasty substances. So odds are mercury and other heavy metal poisoning likely contributed to the archetypical mad-scientist meme immortalized in Mary Shelley's Frankenstein. I doubt it did much for the mental health of several Spanish Lords who proudly bragged they slept on a mercury/water bed either.
Right around the same time unscrupulous alchemists were extracting the last precious coins from the Mafia-like Crime Syndicates who ran feudal Europe, others began to find real uses for quicksilver. One of the first was Evangelista Torricelli, a student of none other than Galileo Galilee. Torricelli's invention is something any red-blooded American kid can understand: I bet every one of you at one time or another trapped liquid in a straw by dipping it in your glass and capping one end with your thumb ... And I bet a bunch of you withdrew that straw, admired the antigravity trick, and then spewed the liquid at your siblings and friends! Ahh the innocence ...
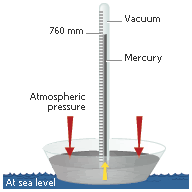
Left: The water trick every kid knows. Right: The finger cap is replaced with a sealed top, the water is replaced with mercury, the plastic straw is replaced with a glass tube: The child's trick becomes a mercury barometer
Well, it turns out that there is a limit of about 33 feet in how much water you can hold that way. Few straws are forty feet long of course so it doesn't affect the run-of-the-mill mischievous kid. But it was a big problem for early miners because they couldn't just suck the water out using a single pipe if it was more than 33 feet underground. The reason you're stuck with that limit is because it takes a column of water that tall to equal the weight of the atmosphere at sea level. Torricelli realized that mercury was more practical to use than water because it was over dozen times heavier and thus it would take a much shorter 'straw' to play around with in the laboratory. And the space at the top of the tube was the first hard vacuum ever created in the laboratory; now called a Torricellian Vacuum.
Today we make marks on that glass 'straw' of mercury and call it a barometer. The barometer measures pressure by how much that column varies from time-to-time as read in inches (Or millimeters) on the tube, or measured using a ruler. The unit most often used now is a millibar; One millibar is defined as one dyne per square inch and the average pressure at sea level is about 1013.25 millibars or 14.7 pounds per square inch. When you see storm meteorlogists Steve Gregory and Jeff Masters quoting a pressure of 950mb for hurricane X, they're talking millibars.
Over the years more uses were found for mercury: As a preservative for lumber, an amalgam for precious metals and fillings, and more recently in wide spread electronics applications. Until a few years ago mercury was also used as a preservative in childhood vaccination formula leading to a fierce debate in the medical community over the role mercury may play in the onset of juvenile autism (The latter is particularly confusing for me: Experts I trust from both sides make what sound like solid arguments and each calls the other guy's talking points total nonsense, on the same level as Young Earth Creationism). Today mercury is used in over 3,000 industrial processes. And therein lays the rub.
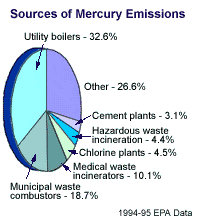

Mercury compounds are released from various industries into the environment where they accumulate at the base of the food chain and become more concentrated as they move up through it
Most incidents of severe mercury poisoning stem from inhalation and ingestion of mercury methylates. And by far most of those pollutants come from the burning of fossil fuels and incineration of materials containing mercury compounds, producing a double whammy for us human critters.
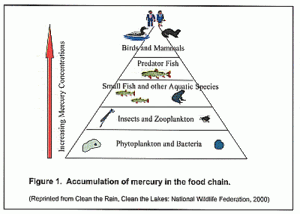
Humans at the top of the food chain ingest the products with the highest accumulations of all
Whether released by fossil fuel or the incineration of waste, the airborne emissions eventually settle onto the surface. There they undergo a dangerous concentration process in plant and animal tissue as they progress up through the food chain, and we directly ingest them via air and water adding to the mercury levels gained from other sources. In particular fish is a source of concern. Humans consume few top land predators. Tiger and bear fillet is a rarity on the dinner menu at DarkSyde Manor. But much of the fish we eat are the large predatory critters at the top of the marine or fresh water food chain, such as Salmon. For this reason fish is of particular concern and mercury levels need to be monitored closely in those items. Some people are more prone to mercury poisoning than others. But anyone who consumes or ingest a relatively small quantity will, sooner or later, exhibit the full adverse consequences of the concentrated mercurial toxins including damage to the brain, liver, kidneys, and just about everything else.
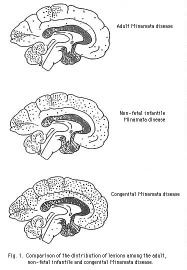
Each tiny dot represents a lesion. The earlier in life and the longer the individual is exposed, the greater the brain damage. This data was first obtained from victims of Minamata Disease: So-named because it was originally diagnosed among residents of Minamata, Japan, in the 1950s after a factory dumped tons of mercury waste into the nearby bay over several decades.
Given the documented and horrific effects of mercury on adults, children, and developing fetuses, starting thousands of years ago and since recorded in gruesome clinical detail, naturally our government is concerned enough to ... buahahahah! Sadly, no.
Until last year methyl mercury emissions were almost unregulated. After an almost ten-year long legal battle waged by the Natural Resources Defense Council, the EPA was finally forced to issue updated mercury emission regulations starting in January of 2004. The new policy was announced with great fanfare by Lawmakers -- with no mention that most of them dragged their feet at every step for decades and were practically forced at gunpoint to comply with the ruling.
Among a number of controversial provisions, the anemic regulations in the so-called Clear Skies Act allow the worst mercury polluters to trade or buy 'mercury credits' with those that do not emit mercury or do not exceed limits mandated by the EPA. The allowable limits are a topic of intense criticism anyway and the usefulness of the trading loophole is questionable; it practically guarantees localized mercury levels well in excess of dangerous amounts. The idea that some far away county is below allowable levels would likely be of little comfort to those suffering from mercury poisoning: Not to mention even these existing rules don't fully kick in for over a decade!
And if that prospect isn't scary enough to jump-start this Halloween Science theme, the tactics used by right-wing shills to deny, obfuscate, and flat out lie about the danger poised by mercury pollution are ghastly in their own right. Science writer Chris Mooney points out in his book The Republican War on Science:
... environmentalists may have shaded the evidence their way, [but] they have hardly concocted a problem from whole cloth. Mercury pollution does present a real and severe threat, especially to infants and children. Compare environmentalists' crimes to the right-wing Wall Street Journal's declaration, apparently on the basis of information from one industry-funded think tank, that there is 'no credible science showing America faces any health threat at all from current fish consumption.' Greens may twist information now and again, but they can hardly match this blatant denial ...
Most of us know elemental mercury from the silvery strip in our childhood friend -- and ticket to getting out of school for a day -- the thermometer. Which brings me back to Mrs. DS and her Alice in Wonderland get-up. It was a precious outfit she put together out of a set of old white drapes with fringe on them and a few assorted dresses that have seemingly 'shrunk' over the years; ahem. She was really excited, like she is every year, to answer the door and hand out doses of glucose in her costume to the all the young ghouls and goblins that come knocking.
But that morning she came down with a fever and by evening she was laying on the couch moaning in delirium, halfway dressed in her cute little ensemble. I spent the night alternating between answering the door to fork over confectionary treats and taking her temperature--which was flirting with 'dash to the ER' levels. Her fever eventually broke and we put our costumes away for another day. But when I was going through them for this next round I found where I had put that thermometer: It was broken in my Halloween pants pocket. And, I have no desire to become what I was going as.
You see, those olden days of industrial drudgery that produced Lewis Carroll and his subterranean Alician fantasies gave rise to many other, much less endearing, realities. Chief among them was mercury poisoning. Clothiers for example used cloth by the bolt-load that was produced in part using mercury nitrate. Before stiffer restrictions came into effect, Felt in particular was sometimes so contaminated with the stuff that some workers and tailors who used it went barking-at-the-moon insane. That included headwear, drape, dress, and suit makers. The slow descent into full blown paranoid lunacy along with the twitchy mercury tremors and shakes among apparel producers and established couturiers of the era was well known in the nascent fashion industry, and a number of colorful idioms crept into everyday language reflecting this. One of those stereotypes even made an appearance in Alice's Looking Glass world. And, had I absorbed enough mercury from that broken thermometer through my skin by way of my baggy green pants pocket, I could have ended up playing the Mad Hatter for more than a single evening on Halloween.
HAPPY HALLOWEEN KOSSACKS!!


