Let us do a thought experiment. Take two identical sets of ingredients consisting of the following: cut up chicken, flour, shortening, leavening, and milk or water, and salt, black pepper, poultry seasoning, and maybe some garlic and a little extra sage.
For the first set of ingredients, boil the chicken in water with the seasonings, then mix up the flour, shortening, water, and leavening. Roll out the dough and cut into squares, drop the squares in the simmering stock, and cook until done. We have just made chicken and dumplings, and it is a wonderful food.
Now, using the second set of ingredients, dredge the chicken pieces in some of the flour with the seasonings in it, then fry them in hot shortening. Make up the same dough that you did for the dumplings, but cut out circles and cook them in a 400 degree oven until done. We have just made fried chicken and biscuits, and it is a wonderful food.
Using identical ingredients we have prepared two completely different looking, tasting, and smelling meals. The only significant difference is the cooking method: the chicken and dumplings were cooked at a rather low (boiling point of water) temperature, and the fried chicken and biscuits were cooked at a much higher temperature. This is the key. The higher temperatures facilitate a browning reaction (actually scores of different ones), whilst the lower temperatures do not supply the activation energy to get these reactions started. (In all fairness, I usually add some onion, celery, and carrot to the stockpot when making chicken broth, but perfectly acceptable chicken and dumplings can be made without these).

No Browning Reaction


With Browning Reactions
There are two general divisions of browning reactions, caramelization and the Maillard reactions. They are related, but the Maillard (may-lard'; the discover was a Frenchman) reactions are more complex. We will start with caramelization.
In this process, sugars (either free sugars or bound up in starches) are heated to high temperatures (around 340 degrees F for sucrose, about 300 degrees F for glucose, and about 220 degrees F for fructose, higher for starch). The reason for the temperature difference is because glucose and fructose are reducing sugars, more reactive chemically than sucrose and much more reactive than starch. Actually, sucrose is converted to glucose and fructose during caramelization, and it is easy to scorch caramel when making it with table sugar. It is a good idea to have a pan of cold water big enough for you to dip the bottom your cooking vessel to slow down the reaction and avoid burning it.
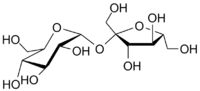
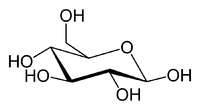
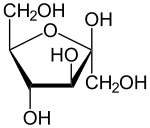
Sucrose Glucose Fructose
So, what happens at these temperatures? The sugars are converted into hundreds of different compounds, some of them much smaller than the sugar molecules, some of them larger. The smaller ones are responsible for much of the fragrances, whilst the larger ones are responsible for the brown color and bitterness in caramel. Since sugar contains only carbon, oxygen, and hydrogen, caramelization products can contain only these elements at most.
Here are some representative aroma compounds formed:
Dicetyl, with a butter scent
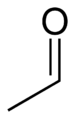
Acetaldehyde, with an odor of sherry in high dilution
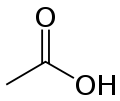
Acetic acid, responsible for the smell of vinegar
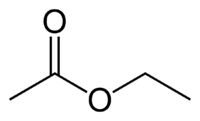
Several esters (ethyl acetate shown here) with fruity notes

Maltol, with a toasty aroma
It should be noted that these fragrances are often unpleasant except in high dilutions. As that sugar continues to be cooked, the blackens and develops a burnt smell, with the residue being high polymers. You can even cook it to the point of carbonizing it.
Remember, caramelization involves only carbohydrates. The other browning reaction, the Maillard reaction, involves the interaction of carbohydrates and amino acids, derived from proteins. There are thousands of variations of the Maillard reaction, and since amino acids all contain nitrogen, and many contain sulfur as well, the resulting range of compounds is much larger than those from caramelization. Interestingly, "caramel" candies are really Maillard candies, since they are the product of sugars and proteins from milk.
The Maillard reaction proceeds at a lower temperature than caramelization of sucrose, at about 250 degrees F. However, it can occur at even lower temperatures as we shall discuss in a bit. In the Maillard reaction, a reducing sugar reacts with an amino acid, either free or in a protein chain. An unstable adduct is formed, which then undergoes further reactions. The result is a complex mixture of flavors and a deep brown color.
Now we see why our chicken and dumplings are completely different than the fried chicken and biscuits. The chicken and dumplings have hardly any Maillard products, whislt both the fried chicken and the biscuits are replete with them. Note also that the flavor of the chicken skin is different when breaded and fried than when broiled without flour. Skin has almost no carbohydrate, so does not undergo Maillard browning but rather a controlled char. Meat has a little carbodydrate in it, and develops a limited amount of Maillard products without breading, but more Maillard products are formed with added carbohydrate. Additionally, some products that also result from Maillard browning can occur from the breakdown of myoglobin, the protein that gives red meat its red color. Bacon, ham, and sausage brown up nicely because of added sugars.
In addition to caramelization products, Maillard flavor families include free amino acids and short chains of amino acids, with a meaty or savory flavor. In addition, one amino acid in particular has a tongue taste as well in contrast to a scent: glutamic acid, formed by a Maillard reaction during the production of soy sauce. This results in soy sauce being a solution of monosodium glutamate, in addition to many other flavors.
Sulfur compounds are largely responsible for the unique taste of carmarelized (actually Maillardized) onions, although some of the sulfur comes from the essential oil that provides the hotness in fresh onions and thus are not strictly Maillard products.
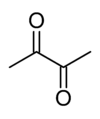
Chocolate gets its color and much of its flavor from Maillard reactions, as does maple syrup and french fried potatoes. In fact, there is one Maillard product that is of some concern in starchy foods that have been cooked at high temperatures. In some potato chips and french fries levels of acrylamide (from the Maillard reaction of the amino acid asparagine and sugars in the potatoes.
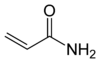
Acrylamide
Sometimes the temperatures do not have to be as high as mentioned provided that other factors are present. For example, dried fruits often turn dark on storage because of Maillard reactions. The high concentration of sugars in dried fruits and the fact that much of that sugar is fructose drive those reactions. Likewise, sweetened condensed milk will turn very dark and develop the taste of milk caramels on storage in warm temperatures. You can actually take advantage of this fact by putting an unopened can of sweetened condensed milk in simmering (not boiling) water for a couple of hours to make an easy caramel pie filling.
The best gravy is made from a roux of flour and some kind of fat, cooked slowly and carefully to develop color and flavor. You can not hurry up a good roux. Some Cajun chefs will cook one for almost an hour, but I think that is getting carried away. Five to seven minutes will produce one completely suitable for gravy. They the liquid (milk, water, or a combination) is stirred in rapidly. With practice, you can stir in the liquid into almost smoking hot roux without getting lumps. I find that a fork is the best tool for this step, although some cooks prefer a whisk. "Gravy" made from raw flour stirred into liquid and then brought to the boil is excellent -- for wallpaper paste. The raw flour taste can not be boiled away in my experience. Even when I make a white sauce, I make a roux with flour and butter and just do not cook it long enough for it to develop much color. For some reason, I find that the addition of salt before cooking the roux improves it, but that might just be superstition on my part. By the way, roux freezes well, so if you make a lot just let it cool and wrap it in polyvinylidine chloride film (Saran or Sam's choice) and freeze. Do not use nonstretch wrap because it is polyethylene and allows oxygen to pass. Use within a couple of months.
There are a few other types of browning, including enzymatic browning of fresh fruit. Cut apples, for example tend to turn brown due to enzyme facilitated oxidation. Antioxidants prevent this, thus using lemon juice or FruitFresh is a preventative. By the way, crushing up a couple of generic Vitamin C tablets does exactly the same thing for much less cost. Fruit Fresh is just Vitamin C dispersed in sugar. At cost of the Vitamin C in that product, your 500 mg Vitamin C tablet would cost almost a dollar rather than the couple of cents you actually pay for it. Enzymatic browning is obviously not usually a desirable process, since fresh fruit should not be brown and these processes harm, rather than enhance, flavor.
Maillard browning reactions are also associated indirectly with food, or not with food at all in some cases. An indirect association is flavoring Bourbon whiskey. By custom and law, Bourbon whiskey is aged in new, charred oak barrels for a minimum period of time. Since wood contains both proteins and sugars, Maillard browning causes the subtle scents to form in the wood that is charred. Aging the fresh whiskey in those barrels causes it to pick up some of the aromas and also some color as well. Bourbon from the still, before aging, is much more like a very rough vodka than what we know as Bourbon. Whilst no one has the definitive answer, one plausible explanation for the discovery of charring the barrels was that only used wine barrels were available to one producer, and he decided to try to burn out the wine color and flavor in them before he aged his whiskey.
Maillard reactions are also to some extent responsible for the color and scent of good compost. Whilst not the only contributor to the constituents of compost, the combination of sugars from the cellulose of the clippings and the proteins in them react over time and with warm temperatures to form the dark color and "earthy" scent of compost.
That about does it for the browning reactions. Any comments are welcome, as are suggestions for recipes, cooking tips, and any other food related information. Tomorrow at 9:00 PM Eastern Pique the Geek will continue the Drugs of Abuse series, this time talking about alcohol. I hope that you can read and comment.
For dinner tonight, I am having Bean Salad. It is a wonderful, easy main course or heavy side. It has been warm the past couple of days, so something cold is attractive. I do not have a definite recipe, but will tell you what I did this time. The common threads are beans, eggs, onion, bell pepper, mayonnaise, salt and pepper, and MSG. I may try putting a few chopped capers in a future batch.
3 cans beans (I used one of Small Red, one of Red Kidney, and one of Black)
1 large green bell pepper, diced
1 medium red onion, diced
3 fifteen minute hard boiled eggs, cooled and diced
Salt to taste
Black pepper to taste
MSG to taste (this recipe really, really needs MSG)
Garlic to taste (garlic powder seems to work best in this recipe)
Mayonnaise to hold together
Drain the beans whilst you dice the pepper and onion and boil and cool the eggs. I like to use canned beans rather than ones that I cook because, if you check the label to assure that they contain calcium chloride they are somewhat firmer than home cooked ones and stand up to mixing. The variety of beans is not that important, but I like to use at least part dark red kidney beans. Be sure to check the label that they do not have sugar added to them. This salad does not like sweet.
Put the onion and pepper into a large bowl and stir in several tablespoons of mayonnaise, and some salt, pepper, and MSG. Go easy with all of these, because you can add more later. Dice the eggs after they are cool, and add them and the drained beans to the mixture in the bowl. Fold gently so as not to bruise the beans and tear up the diced eggs. Taste and adjust seasonings and mayonnaise if needed. Put in a tight container and refrigerate overnight. Like many bean dishes, this one is much improved by allowing the flavors to meld.
Serve cold.
Here is what a bowl of it looks like after refrigeration:

If Ek will provide the current What's for Dinner? calendar in a comment, I will add it here.
UPDATE: Folks, I am tired and need to eat (posting at 7:30 PM sort of knocks me out of dinner). I will check comments tomorrow and respond if appropriate. Thank everyone for posting your views and questions. Once again, do not forget Pique the Geek tomorrow here at 9:00 PM. Thanks for reading!
Warmest regards,
Doc


