You can't buy a lithium-air battery (yet). You've probably never even heard of one. They were only invented 15 years ago, and for now, they're still just laboratory curiosities.
But with the latest lab breakthrough, the lithium-air battery (also known as the lithium-oxygen battery) is nosing up into the energy density region seen in gasoline. If you're thinking battery-powered car, maybe your sights are too low. How does battery-powered airplane sound?
The technical stuff
Batteries are composed of three layers: two electrodes (a negative one called an anode and a positive one called a cathode) separated by an electrolyte. The electrolyte allows cations (atoms or molecules with a positive electric charge, because they're missing an electron) to flow through from anode to cathode, but it won't allow the matching electrons to flow through. So to balance out the charges, the electrons have to take another path from anode to cathode, which is a wire, where they do work in the process. The whole thing works until the electrolyte runs out of ions, at which point the battery has to be recharged.
The lithium-air battery uses metallic lithium as the anode, and air-permeable carbon as the cathode. The electrolyte is a thin gel that allows lithium ions (Li+) from the anode to migrate over to the cathode, where they combine with oxygen from the air to form lithium oxide or lithium peroxide. Eventually one of two things happens: either all the lithium in the anode gets used up, or (more likely) the carbon in the cathode becomes full of lithium oxide and doesn't have room for any more. When the battery is recharged, the lithium oxide is dissociated and the lithium ions migrate back to the anode.
Since lithium is the lightest metal (in weight), any battery using lithium will be light. But since lithium-air batteries also have a light cathode, they are the lightest batteries known.
What's New
Now researchers at MIT have added carbon nanotubes to the cathode, with spectacular results. With a lot more surface area, the cathode can now hold a lot more lithium oxide, which means that the battery can run a lot longer before it needs recharging. The result is four times the energy density of the previous Li-air record holder: the old record of about 600 Watt-hours per kilogram has been upped to about 2500 Wh/kg, in the fully discharged state. This is getting close to the theoretical maximum is 3215 Wh/kg for a discharged lithium-air battery.
Specifying the charge state is important when you talk about the energy density of lithium-air batteries, because the battery actually gains weight as it discharges (since it's taking in oxygen from the air). In the fully-charged state, the battery weighs less: the MIT researchers have reported energy densities of over 10,000 Wh/kg when fully charged, which is essentially at the theoretical limit right now.
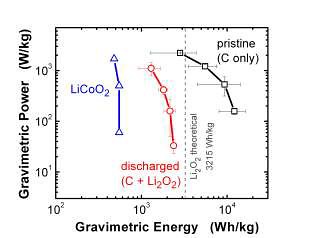
Power-to-weight and energy-to-weight ratios. Blue: previously reported best lithium-air battery with cobalt catalyst cathode (discharged). Red: new MIT battery with carbon nanotube cathode (discharged). Black: new battery fully charged.
Doing a little unit conversion, in the discharged state 2500 Wh/kg = 9 Megajoules per kg, and in the charged state, 11000 Wh/kg = 40 MJ/kg. For comparison, gasoline is about 44 to 47 MJ/kg, and jet fuel is 43. So in the fully charged state, the Li-air battery is close to the fossil fuel level. (The lithium-ion battery in your cell phone is around 1 MJ/kg, the alkaline battery in your flashlight is 0.6 MJ/kg, and the lead-acid battery that starts your car is about 0.1 MJ/kg. So even in its discharged state, 9 MJ/kg is pretty huge.)
The implications
This has obvious implications for the transportation industry, where the weight (and cost, and availability) of batteries is the biggest obstacle to practical electric cars. Note that the power-to-weight ratio reported is between 1000 and 2000 W/kg, or between 0.6 and 1.2 hp per lb, which also compares favorably to a gasoline engine.
But when you consider a battery this energy dense, air travel also presents an interesting opportunity. Imagine an aircraft with a hybrid engine that uses both battery power from a Li-air battery, and energy from burning renewable biofuel. As the plane flies, the liquid fuel runs out and the plane gets lighter; but the battery takes on oxygen, and the plane gets heavier. So it would be possible to design a hybrid-powered aircraft that stays at the same weight throughout the entire flight, as weight lost and weight gained were exactly counterbalanced.
Currently, as a plane flies it loses weight, and to compensate the pilot must lose lift in an equal amount, which he or she does by pointing the nose just slightly more downward as the flight proceeds. This affects what's called the "angle of attack" of the wing and reduces lift (and also reduces drag). But while lift varies linearly with angle of attack, drag varies exponentially, which means that there is one and only one angle of attack at which the ratio of lift-to-drag is at it's highest. At this precise angle of attack, the plane is operating at its most efficient point, aerodynamically speaking.
Pilots today don't worry about this efficient point at all (except for a few long-distance record setters). That's because pilots know they can only fly at that ideal angle of attack for a short while before fuel burns off, lift must be reduced, and the angle of attack must be changed to something less ideal. But with a constant-weight airplane, it would be possible to fly the airplane at the ideal angle of attack for the entire flight, making the entire trip more energy efficient. All a pilot would have to do would be to know the weight of the aircraft when loaded, then look up the correct airspeed for weight and altitude on a chart, and fly at that airspeed.
Links:
Peer-reviewed abstract in Energy & Environmental Science
MIT press release
Supplemental data (pdf)


