For those familiar with the devastating agony of losing their cognitive and emotional capacities, or those who are losing a loved one to such a painful neurodegenerative brain disease, this breakthrough in the creation of a new type of cells derived from stem cells, and described in New stem-cell-derived cells hold promise for Alzheimer’s, other brain diseases, offers hope.
Scientist from UC Irvine have developed a new kind of cell called choroid plexus epithelial cells (CPECs) from mouse and human embryonic stem cell lines which show promise in repairing and rejuvenating the brain's ability to produce cerebrospinal fluid (CSF) that is responsible for removing metabolic waste and foreign substances from our brains and spinal chords. Insufficiency and dysfunction of the choroid plexus is suspected in playing a large role in many neurodegenerative diseases.
In neurodegenerative diseases, the choroid plexus and CPECs age prematurely, resulting in reduced CSF formation and decreased ability to flush out the plaque-forming proteins that are a hallmark of Alzheimer’s. Transplant studies have provided proof of concept for CPEC-based therapies. However, such therapies have been hindered by the inability to expand or generate CPECs in culture.
Here we can better understand how CSF help "flush out the plague-forming proteins," thereby improving brain function, by imagining how an energized and broad alliance of progressive voters flushes out damaging Tea Party and Republican obstructionist congressmen, thereby improving the functioning of congress. In this heuristic metaphor, choroid plexus epithelial cells function like progressive internet bloggers, like those of us here at Daily Kos, where we help to produce more CSF-like high information progressive voters and GOTV volunteers.
Here's one of those cute kind of diagrams that scientist put in their articles thinking that it helps people understand their work, and also it illustrates how tolerating articles like this on a political blog directly contributes to electing more and better Democrats. I include it here for a humorous comic interlude before continuing below the fleur de orange.
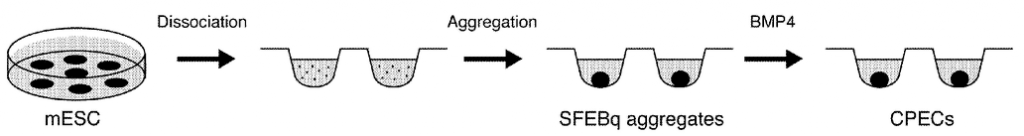 Choroid plexus epithelial cells generated in a culture medium using embryonic stem cells
Choroid plexus epithelial cells generated in a culture medium using embryonic stem cells
(credit: Edwin S. Monuki and Momoko Watanabe/USPTO)
If I had better graphics software I would put a "You are here" arrow pointing to BMP4 on this diagram to illustrate how bone morphogenetic protein (BMP) signaling induces the differentiation of CPEC cells in a way that is metaphorically similar to the impact that reading articles like this has on differentiating more highly educated and pro-Science Kossacks ready to "wash those conservatives right out of our congress."
This breakthrough could open the way for inexpensive mass production of this kinds of epithelial cells which could then be transplanted into the brains of those experiencing age related loss, thereby keeping brains healthier in case we should decide to use them later.
“Our method is promising, because for the first time we can use stem cells to create large amounts of these epithelial cells, which could be utilized in different ways to treat neurodegenerative diseases,” said Monuki, an associate professor of pathology & laboratory medicine and developmental & cell biology at UCI. ...
These cells could be part of neurodegenerative disease treatments in at least three ways, Monuki said. First, they’re able to increase the production of CSF to help flush out plaque-causing proteins from brain tissue and limit disease progression. Second, CPEC “superpumps” could be designed to transport high levels of therapeutic compounds to the CSF, brain and spinal cord. Third, these cells can be used to screen and optimize drugs that improve choroid plexus function.
As next steps the authors plan to use these same CPECs to test the affect in mice of Huntington's, Alzheimer's and pediatric diseases.
It is worthy of note that this research was funded in part by a grant from the National Institutes of Health, along with the California Institute for Regenerative Medicine, UCI’s Institute for Clinical & Translational Science, and UCI’s Alzheimer’s Disease Research Center, and that the NIH is one of examples of "bloated" government spending the Mitt Romney, Paul Ryan, and his deficit hawk Republicans want to cut.
One reason I'm proud to support the Democratic Party, and also publish articles such as this, that may not appear relevant on a political blog, is to illustrate the distinction between us pro-science, and pro-research Democrats who see value in investing in science, medicine, and the whole idea of creating a better future for all Americans, and the Republican Party and Paul Ryan who are anti-science and advocate cutting funding for the NIH, NFA, NSF, EPA, and virtually every component of government spending except defense.
It makes a difference. We should strongly support returning the tax rate on the top bracket from 34% to 39%, and closing tax loopholes in order to sustain incredibly valuable research like this, and keep these kinds of scientists and science in the US.
For those of you who would like to see more articles like this please follow me at HoundDog by supportively clicking the little heart by my name.
*** end of article ****
For those of your with greater interest here is a link to the article in the Journal of Neuroscience:
BMP4 Sufficiency to Induce Choroid Plexus Epithelial Fate from Embryonic Stem Cell-Derived Neuroepithelial Progenitors by Momoko Watanabe1, Young-Jin Kang2, Lauren M. Davies2, Sanket Meghpara2, Kimbley Lau2, Chi-Yeh Chung2, Jaymin Kathiriya2, Anna-Katerina Hadjantonakis3, and Edwin S. Monuki1,2,4
Abstract
Choroid plexus epithelial cells (CPECs) have essential developmental and homeostatic roles related to the CSF and blood–CSF barrier they produce. Accordingly, CPEC dysfunction has been implicated in many neurological disorders, such as Alzheimer's disease, and transplant studies have provided proof-of-concept for CPEC-based therapies. However, such therapies have been hindered by the inability to expand or generate CPECs in culture. During development, CPECs differentiate from preneurogenic neuroepithelial cells and require bone morphogenetic protein (BMP) signaling, but whether BMPs suffice for CPEC induction is unknown. Here we provide evidence for BMP4 sufficiency to induce CPEC fate from neural progenitors derived from mouse embryonic stem cells (ESCs). CPEC specification by BMP4 was restricted to an early time period after neural induction in culture, with peak CPEC competency correlating to neuroepithelial cells rather than radial glia. In addition to molecular, cellular, and ultrastructural criteria, derived CPECs (dCPECs) had functions that were indistinguishable from primary CPECs, including self-assembly into secretory vesicles and integration into endogenous choroid plexus epithelium following intraventricular injection. We then used BMP4 to generate dCPECs from human ESC-derived neuroepithelial cells. These findings demonstrate BMP4 sufficiency to instruct CPEC fate, expand the repertoire of stem cell-derived neural derivatives in culture, and herald dCPEC-based therapeutic applications aimed at the unique interface between blood, CSF, and brain governed by CPECs.
And, for those interested here is a link to the patent application:
Patent application title: GENERATION OF CHOROID PLEXUS EPITHELIAL CELLS FROM HUMAN EMBRYONIC STEM CELLS
Inventors: Edwin S. Monuki (Irvine, CA, US) Momoko Watanabe (Irvine, CA, US)
IPC8 Class: AA61K3530FI
USPC Class: 424 937
Class name: Drug, bio-affecting and body treating compositions whole live micro-organism, cell, or virus containing animal or plant cell
Publication date: 2012-08-09
Patent application number: 20120201789
Abstract:
Choroid plexus epithelial cells are generated in a culture medium using embryonic stem cells and adding an effective amount of bone morphogenetic protein and/or other members of the transforming growth factor beta (TGF-beta) superfamily. Generation of such choroid plexus epithelial cells are confirmed using a combination of genetic markers, antibodies, histology inspection, functional assays, and integration into the endogenous choroid plexus in mice.
Claims:
1. A method of inducing choroid plexus epithelial cells in vitro comprising providing embryonic stem cells; and adding an effective amount of bone morphogenetic protein and/or other members of the transforming growth factor beta (TGF-beta) superfamily.
Read more: http://www.faqs.org/...


