When our son was in kindergarten, we took him to see the Grateful Dead in Soldier Field. On the way to the show we passed a street vender selling small balloons. It led to an awkward explanation about why he cannot have one of those balloons. How do you explain nitrous oxide to a kid? His response was "Really?!?!?"
What jogged this memory was a study just published in the Proceedings of the National Academy of Sciences about nitrous oxide (N2O). This is a very dangerous whippet for the climate.
According to the EPA:
Nitrous oxide molecules stay in the atmosphere for an average of 120 years before being removed by a sink or destroyed through chemical reactions. The impact of 1 pound of N2O on warming the atmosphere is over 300 times that of 1 pound of carbon dioxide.
It sounds like we should not dump much of this stuff into the atmosphere. One major source of N2O emissions is from soils, particularly those with treated with synthetic fertilizers. In the words of study authors Iurii Shcherbak, Neville Millar, and G. Philip Robertson:
Nitrous oxide (N2O) is a potent greenhouse gas (GHG) that also depletes stratospheric ozone. Nitrogen (N) fertilizer rate is the best single predictor of N2O emissions from agricultural soils, which are responsible for ∼50% of the total global anthropogenic flux, but it is a relatively imprecise estimator.
Dumping too much synthetic fertilizer into the soil is stupid. So here is the question. The Intergovernmental Panel on Climate Change (IPCC) has always assumed a linear relationship between the dose (concentration of synthetic nitrogen in fertilizer applied to fields) and the response (N2O emissions from the soil). It is a reasonable starting point when formulating models, but does it provide the best fit to real-world data?
The study from the Michigan State researchers provided compelling evidence that the relationship between fertilizer dose and soil emissions response is likely to be nonlinear. Combining data from all published studies:
We found that the N2O response to N inputs grew significantly faster than linear for synthetic fertilizers and for most crop types. Our results suggest a general trend of exponentially increasing N2O emissions as N inputs increase to exceed crop needs. Use of this knowledge in GHG inventories should improve assessments of fertilizer-derived N2O emissions, help address disparities in the global N2O budget, and refine the accuracy of N2O mitigation protocols.
This graph illustrates the departure from IPCC model assumptions for N2O relative to available data in published studies.
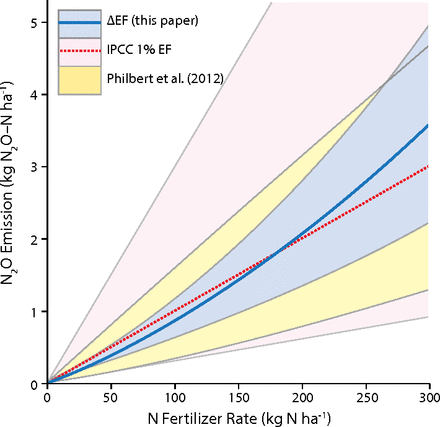
Comparison of the uncertainties associated with IPCC tier 1 (1%), a range of six models from Philibert et al. (31), and the mean ∆EF model for all site-years from this metaanalysis (excluding N-fixing crops and the bare soil site-year). The 95% CI is provided for each model across a range of N fertilizer rates (0–300 kg·ha−1). The IPCC tier 1 95% CI is 0.3–3%. The Philibert et al. (31) 95% CI encompasses parameter uncertainty.
That means that industrial scale farms that apply large amounts of synthetic fertilizer to their fields will have a much larger impact on climate change than originally feared. Brilliant. The data compiled by this study can serve as a good starting point for empirically-derived maximum concentrations to prevent N inputs far in excess of crop demands. Of course that requires meaningful agricultural policy, one not dictated by agribusiness corporations. Pardon me if I do not hold my breath waiting for sensible regulation to protect the environment from nutrient pollution and its climate consequences.


